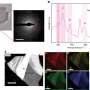
Cave of Crystals, Naica, Mexico. Image credit: Skylar
(PhysOrg.com) -- Deep within a silver and lead mine in Naica, Mexico, scientists discovered what is now known as Cueva de los Cristales, or Cave of Crystals, close to a decade ago. The gypsum crystals found in this cave measure as long as 11 meters (36 feet) and as thick as 1 meter (3 feet). While these crystals are beautiful and reminiscent of a Superman film, they have had scientists stumped since their discovery. The question was just how long these crystals had been growing in order to become this large.
Because growing a giant crystal in a laboratory creates difficulties because of the timescale required, they were unable to make an accurate guess as to the age of these crystals. However, thanks to a team of researchers from Spain and Japan and a new analytical technique, the age and the growth rate of these crystals has been determined and they have been a long time in the making. As much as 1 million years in the making. The researchers study has been published in the September 12 online version of the Proceedings of the National Academy of Sciences.
The new technique utilizes a beam of white light aimed at the crystal to determine its properties. Scientists determined that the gypsum crystalized within the cave at temperatures between 54 and 58 degrees Celsius, or 129 to 136 degrees Fahrenheit. By submersing the crystal in mineral rich water, similar to what would have flowed in the cave before it was drained in 1975, the researchers discovered that the slowest growth would have occurred when the cave was 55 degrees Celsius. At this temperature, the crystals would have taken 990,000 years to form with a diameter of 1 meter. By increasing the temperature in the cave by one degree, or 56 degrees Celsius, the same size crystal could have formed in a little less than half the time, or around 500,000 years. This possible growth rate would work out to around a billionth of a meter of growth per day and is the slowest growth rate that has ever been measured.
Time course of the surface morphology of the {010} face of a gypsum crystal in contact with Milli-Q water at room temperature (22.5 C). Movie: PNAS, doi: 10.1073/pnas.1105233108
After determining that these crystals could very well be close to a million years old, the researchers are now hoping to look for microscopic liquid pockets within the crystals in the hopes of finding possible microorganisms inside.
More information: Ultraslow growth rates of giant gypsum crystals, PNAS, Published online before print September 12, 2011, doi:10.1073/pnas.1105233108
Abstract
Mineralogical processes taking place close to equilibrium, or with very slow kinetics, are difficult to quantify precisely. The determination of ultraslow dissolution/precipitation rates would reveal characteristic timing associated with these processes that are important at geological scale. We have designed an advanced high-resolution white-beam phase-shift interferometry microscope to measure growth rates of crystals at very low supersaturation values. To test this technique, we have selected the giant gypsum crystals of Naica ore mines in Chihuahua, Mexico, a challenging subject in mineral formation. They are thought to form by a self-feeding mechanism driven by solution-mediated anhydrite-gypsum phase transition, and therefore they must be the result of an extremely slow crystallization process close to equilibrium. To calculate the formation time of these crystals we have measured the growth rates of the {010} face of gypsum growing from current Naica waters at different temperatures. The slowest measurable growth rate was found at 55 °C, 1.4 ± 0.2 × 10-5 nm/s, the slowest directly measured normal growth rate for any crystal growth process. At higher temperatures, growth rates increase exponentially because of decreasing gypsum solubility and higher kinetic coefficient. At 50 °C neither growth nor dissolution was observed indicating that growth of giant crystals of gypsum occurred at Naica between 58 °C (gypsum/anhydrite transition temperature) and the current temperature of Naica waters, confirming formation temperatures determined from fluid inclusion studies. Our results demonstrate the usefulness of applying advanced optical techniques in laboratory experiments to gain a better understanding of crystal growth processes occurring at a geological timescale.
© 2011 PhysOrg.com