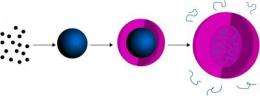
This is a schematic diagram of the approach used by chemists at Georgia Tech to synthesize poly(N-isopropylmethacrylamide) semi-hollow microgel capsules containing a low density of trapped poly(N-isopropylacrylamide) chains. The capsule's multiple-compartment structure makes it a good candidate for simultaneously delivering hydrophobic and hydrophilic drugs into the body. Credit: Georgia Tech/L. Andrew Lyon
Researchers at the Georgia Institute of Technology have designed a multiple-compartment gel capsule that could be used to simultaneously deliver drugs of different types. The researchers used a simple "one-pot" method to prepare the hydrogel capsules, which measure less than one micron.
The capsule's structure -- hollow except for polymer chains tethered to the interior of the shell -- provides spatially-segregated compartments that make it a good candidate for multi-drug encapsulation and release strategies. The microcapsule could be used to simultaneously deliver distinct drugs by filling the core of the capsule with hydrophilic drugs and trapping hydrophobic drugs within nanoparticles assembled from the polymer chains.
"We have demonstrated that we can make a fairly complex multi-component delivery vehicle using a relatively straightforward and scalable synthesis," said L. Andrew Lyon, a professor in the School of Chemistry and Biochemistry at Georgia Tech. "Additional research will need to be conducted to determine how they would best be loaded, delivered and triggered to release the drugs."
Details of the microcapsule synthesis procedure were published online on July 5, 2011 in the journal Macromolecular Rapid Communications.
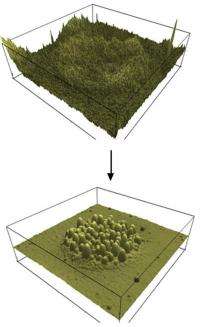
These three-dimensional atomic force microscopy images show the formation of microcapsules with nanoscale inclusions when the temperature is increased. The multiple-compartment microcapsules could be used to simultaneously deliver hydrophobic and hydrophilic drugs into the body. Credit: Georgia Tech/L. Andrew Lyon
Lyon and Xiaobo Hu, a former visiting scholar at Georgia Tech, created the microcapsules. As a graduate student at the Research Institute of Materials Science at the South China University of Technology, Hu is co-advised by Lyon and Zhen Tong of the South China University of Technology. Funding for this research was provided to Hu by the China Scholarship Council.
The researchers began the two-step, one-pot synthesis procedure by forming core particles from a temperature-sensitive polymer called poly(N-isopropylacrylamide). To create a dissolvable core, they formed polymer chains from the particles without a cross-linking agent. This resulted in an aggregated collection of polymer chains with temperature-dependent stability.
"The polymer comprising the core particles is known for undergoing chain transfer reactions that add cross-linking points without the presence of a cross-linking agent, so we initiated the polymerization using a redox method with ammonium persulfate and N,N,N',N'-tetramethylethylenediamine. This ensured those side chain transfer reactions did not occur, which allowed us to create a truly dissolvable core," explained Lyon.
This is an atomic force microscopy (top) height image and (bottom) phase image of microcapsules synthesized by L. Andrew Lyon, a professor in the School of Chemistry and Biochemistry at Georgia Tech. The multiple-compartment gel capsules could be used to simultaneously deliver drugs of different types. Credit: Georgia Tech/L. Andrew Lyon
For the second step in the procedure, Lyon and Hu added a cross-linking agent to a polymer called poly(N-isopropylmethacrylamide) to create a shell around the aggregated polymer chains. The researchers conducted this step under conditions that would allow any core-associated polymer chains that interacted with the shell during synthesis to undergo chain transfer and become grafted to the interior of the shell.
Cooling the microcapsule exploited the temperature-sensitivities of the polymers. The shell swelled with water and expanded to its stable size, while the free-floating polymer chains in the center of the capsule diffused out of the core, leaving behind an empty space. Any chains that stuck to the shell during its synthesis remained. Because the chains control the interaction between the particles they store and their surroundings, the tethered chains can act as hydrophobic drug carriers.
Compared to delivering a single drug, co-delivery of multiple drugs has several potential advantages, including synergistic effects, suppressed drug resistance and the ability to tune the relative dosage of various drugs. The future optimization of these microcapsules may allow simultaneous delivery of distinct classes of drugs for the treatment of diseases like cancer, which is often treated using combination chemotherapy.
Provided by Georgia Institute of Technology