Scientists generate two energetic electronic states from one photon
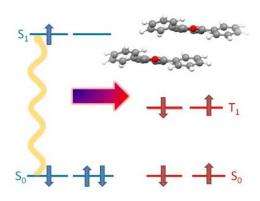
(PhysOrg.com) -- Researchers from the U.S. Department of Energy's National Renewable Energy Laboratory (NREL) and the University of Colorado, Boulder (UCB), have reported the first designed molecular system that produces two triplet states from an excited singlet state of a molecule, with essentially perfect efficiency. The breakthrough could lead to a 35 percent increase in light-harvesting yield in cells for photovoltaics and solar fuels. The experiments, using a process called singlet fission, demonstrated a 200 percent quantum yield for the creation of two triplets of the molecule 1,3-diphenylisobenzofuran (DPIBF) at low temperatures. The research has been published in the Journal of the American Chemical Society.
Under a collaborative research program sponsored by the U.S. Department of Energy, scientists at NREL and the University of Colorado, Boulder report the first designed molecular system that produces two triplet states from an excited singlet state of a molecule (1,3-diphenylisobenzofuran (DPIBF)) with essentially perfect efficiency. The process is called singlet fission, and the experiments indicated a quantum yield at 77K of 200% for the creation of the two triplets in DPIBF. This means that it may be possible to generate two electron-hole pairs from absorbed single photons in solar cells based on singlet fission, and thus increase its conversion efficiency into electricity or solar fuels by 35%.
The research has been published in the Journal of the American Chemical Society in the online ASAP version prior to final publication; the manuscript is entitled “High triplet yield from singlet fission in a thin film of 1,3-diphenylisobenzofuran.” The NREL authors are Justin C. Johnson, Sr. Scientist in the Center for Chemical and Materials Science, and Arthur J. Nozik, Sr. Research Fellow; the author at the University of Colorado is Professor Josef Michl, who heads a team of graduate students, post docs, and several other collaborators working on singlet fission.
Singlet fission is the molecular analogue of Multiple Exciton Generation (MEG) in inorganic semiconductor quantum dots; MEG is another process under investigation at NREL whereby two electron-hole pairs, existing as excitons in quantum dots, can be generated from single photons. Thermodynamic modeling indicates that a relatively simple bilayer solar cell incorporating singlet fission (or MEG) could increase the photoconversion yield of an ideal solar cell by more than one-third.
Both the molecular design principles and broader concepts of coupling chromophores into functional networks were key advances that produced 2 electrons per absorbed photon for future use in cells for photovoltaics and solar fuels. The NREL research on singlet fission was sponsored by the Hydrogen Fuel Initiative within DOE’s Office of Science, Office of Basic Energy Sciences, Division of Chemical Sciences, Biosciences, and Geosciences. Professor Michl’s research was funded by the Photovoltaics Program at the DOE Office of Energy Efficiency and Renewable Energy.
Until this recent advance, singlet fission had been known as a somewhat obscure phenomenon occurring primarily in single crystals of tetracene, and most of the investigations occurred more than three decades ago. Recent notions of utilizing singlet fission in light harvesting schemes were developed by Hanna and Nozik (Hanna, M. C.; Nozik, A. J. J. Appl. Phys. 2006, 100, 074510/1), leading to a much more extensive search for appropriate candidate molecules. However, in the absence of detailed mechanistic information about the process itself it was unclear about how to proceed.
The compound DPIBF was identified as a promising candidate by using theory to direct a search for chromophores likely to have a desirable ratio of singlet and triplet excitation energies (Paci, I. et al J. Amer. Chem. Soc. 2006, 128, 16546), the latter of which can be difficult to measure experimentally. Fundamental characterization of the DPIBF molecules isolated in solution (Schwerin, A. et al J. Phys. Chem. A 2010, 114, 1457-1473) was followed up by considerations of how best to connect two DPIBF molecules together (Greyson et al, J. Phys. Chem B ASAP), a necessary step in order to have two triplets form after photoexcitation. Initial attempts to couple the molecules covalently in a chain-like fashion did not produce high singlet fission yields (Michl et al Proc. SPIE 2007, 6656, 66560E1), but more recent theory (Smith et al. Chem. Rev. in press) has led to the discovery that the natural geometry that the molecules adopt while packing into crystals, a sandwich type orientation, might be optimal.
Due to the fact that single bulk crystals can be disadvantageous for optical spectroscopy, thin polycrystalline films of DPIBF were fabricated by subliming the compound in vacuum onto prepared substrates. The details of the process of forming these films are extremely important to the ultimate result because the nanoscale molecular geometry depends on the nature of the microcrystals that form. When done correctly, the films showed strong spectroscopic signatures indicating a high yield of triplets. In order to quantify the yield, ultrafast laser experiments were performed to accurately measure the initial populations of photoexcited singlet states and their time evolution into triplets on a picosecond time scale.
A careful analysis of the data led to yields approaching 200% and an increase in the triplet formation rate of more than ten thousand compared with the isolated DPIBF molecule. Both of these observations support the notion that efficient singlet fission is occurring and that the design criteria set forth may be quite general.
Further work on DPIBF and compounds that absorb a larger fraction of the solar spectrum could lead to a significant future role for singlet fission chromophores in various types of ultraefficient light harvesting devices.
More information: Paper online: pubs.acs.org/doi/abs/10.1021/ja104123r
Provided by National Renewable Energy Laboratory