Long-distance communication, microbial style

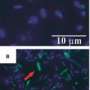
Scientists knew that the microbe Shewanella oneidensis transformed the electronic structure of the iron oxide it touched in the ground as well as without direct contact. Scientists from Pacific Northwest National Laboratory have now developed a model system that allows the study of this indirect transformation. They also discovered previously unseen iron(II) phosphate rosettes precipitated during bioreduction process. These blooms grew on the surface and within the grains of sand and were intensely colonized by the microbes.
This research provides a greater understanding of redox transformations of iron(III) hydroxides, which influence the fate of pollutants and nutrients in certain soils, sediments, and subsurface materials. "These findings were both fascinating and unexpected," said Dr. Tanya Peretyazhko, who led the PNNL team.
To do this study, the team devised a new model system that mimics the real world where iron oxides are commonly embedded in microscopic pores between grains of sand and other matter or in microfractures in the subsurface. The team simulated intragrain pores and microfractures by mixing a solution of iron oxide nanoparticles with highly porous silica grains. They tested the grains to verify that when in contact with water, the iron oxides would remain embedded within the pores.
Then, the S. oneidensis was introduced. Bioreduction was encouraged in oxygen-free conditions and with the introduction of chemicals, such as phosphate. With ideal conditions established, the team developed techniques to preserve the samples for x-ray diffraction and scanning and transmission electron microscopy, which were performed in EMSL, a Department of Energy national scientific user facility at PNNL. This analysis answered questions about shape and mineral composition of iron precipitates and the proximity of the microbes.
With the simulation technique established, the team studied the mechanism that microbes use to transform iron oxides without direct contact, as well as mechanisms of precipitate formation.
The researchers are now looking to understand how the microbes accomplish their long-distance electron transfer. Some researchers suggest "electron shuttles," in which microbes release molecules that carry electrons to the iron oxides. Others posit that organic molecules are released to the iron oxides and dissolve them without reduction.
More information: Peretyazhko TS, JM Zachara, DW Kennedy, JK Fredrickson, BW Arey, JP McKinley, CM Wang, AC Dohnalkova, and Y Xia. 2010. "Ferrous phosphate surface precipitates resulting from the reduction of intragrain 6-line ferrihydrite by Shewanella oneidensis MR-1." Geochimica et Cosmochimica Acta 74(12):3751-3767.
Provided by Pacific Northwest National Laboratory