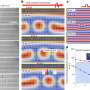
Cellular mechanical forces may initiate angiogenesis
Pericytes, the contractile cells surrounding capillaries, may use mechanical forces to initiate angiogenesis, the "sprouting" of new blood vessels, according to researchers at Tufts University School of Medicine (TUSM) and the Massachusetts Institute of Technology (MIT). The study, published in Journal of Physics: Condensed Matter, is among the first to examine mechanical signaling by pericytes as a potential driver of angiogenesis, which is crucial in the progression of cancer, diabetic retinopathy, and age-related macular degeneration.
Previously, scientists have focused on the chemical signals that initiate angiogenesis and have developed drugs to alter these signals. Now, it appears that the mechanical signals from pericytes can also play a role in regulating the sprouting of new blood vessels.
"If we find that mechanical signaling, such as the force exerted by pericytes, initiates angiogenesis, we can explore ways of interfering with these signals. In this in vitro study, we found that pericytes generated contractions that physically altered the microvascular environment. In the body, such local contractions could serve as the initiating, mechanical signals that influence angiogenesis," said co-senior author Ira Herman, PhD, professor of physiology at TUSM.
"Depending on the circumstances, angiogenesis can either promote health or promote disease," he continued. "Angiogenesis is required for early development and wound healing, but it can also feed cancerous tumors or cause vascular complications in the eye. Our goals are aimed at developing drugs that might enhance wound healing angiogenesis while preventing the harmful angiogenesis that leads to tumor growth or vision loss."
Herman and team members at Tufts joined materials scientists and engineers at MIT in this interdisciplinary effort.
"This collaboration enabled us to quantify the role that mechanics plays in angiogenesis at the cellular level. We directly imaged and quantified the contraction these pericytes exerted to wrinkle the underlying membrane and examined how specific drugs amplified and mitigated this contractile force. These measurements allowed us to estimate how much pericytes contracted and stiffened the microvascular environment, sending mechanical signals to nearby cells," said co-senior author Krystyn J. Van Vliet, PhD, associate professor of materials science and engineering at MIT.
The researchers isolated pericytes, using criteria Herman helped develop, and applied them to a silicone membrane. With an atomic force microscope, researchers at MIT measured the stiffness of the contracting pericytes and the consequent degree of wrinkling the pericytes caused in the membrane. Pericytes generated contractions that caused underlying membranes to shorten by an average of 38 percent.
The first authors of the study are Sunyoung Lee, PhD, and Adam Zeiger, a PhD candidate, both members of the laboratory for material chemomechanics in the department of materials science and engineering at MIT. Additional authors are John Maloney, a PhD candidate and member of Van Vliet's laboratory for material chemomechanics in the department of materials science and engineering at MIT, and Maciej Kotecki, MD, PhD, a research associate in Herman's laboratory in the department of physiology at TUSM.
Krystyn Van Vliet is also an associate professor in the department of biological engineering at MIT. Ira Herman is also a member of the faculty in cell, molecular and developmental biology and in the cell and molecular physiology program at the Sackler School of Graduate Biomedical Sciences at Tufts.
More information: Lee S, Zeiger A, Maloney J, Kotecki M, Van Vliet KJ, Herman IM. Journal of Physics: Condensed Matter. 2010. 22 (194115). "Pericyte actomyosin-mediated contraction at the cell-material interface can modulate the microvascular niche." Published online April 26, 2010, doi: 10.1088/0953-8984/22/19/194115
Provided by Tufts University