Researchers program cells to be remote-controlled by light
(PhysOrg.com) -- UCSF researchers have genetically encoded mouse cells to respond to light, creating cells that can be trained to follow a light beam or stop on command like microscopic robots.
This is the first time researchers have been able to import a light controlled "on-off switch" from plants into a mammalian cell to instantly control a variety of cell functions, the researchers said. As such, it offers both a powerful new tool in cancer and cardiovascular research, as well as the potential to ultimately control complex processes such as nerve growth.
The findings appear in the September 13 advanced online publication of the journal "Nature" and are available at nature.com. They are reported alongside a paper on similar research led by Klaus Hahn, Ph.D., and his colleagues at the University of North Carolina, Chapel Hill.
Together, the papers are the first to demonstrate that plant light-switches can be imported into mammalian cells to control complex regulatory processes. The UCSF research is unique in developing a generic plug-and-play switch, based on protein recruitment, which can be wired to control diverse processes in many types of cells and organisms, the researchers said.
The findings could have various therapeutic applications down the road, such as the ability to guide nerve cells to reconnect across a broken spinal pathway in a spinal cord injury, according to Wendell Lim, PhD, one of three senior authors on the paper and the director of the Cell Propulsion Laboratory, a National Institutes of Health Nanomedicine Development Center at UCSF and UC Berkeley.
More immediately, the findings offer a new approach for scientific research into the complex regulatory processes involved in diseases like cancer and inflammation, he said.
"This is a powerful tool for cell biology and cancer research," said Lim, who is a professor in the UCSF Department of Cellular and Molecular Pharmacology. "If you have a controllable 'light switch' that is generic enough to use in multiple cell functions, it gives you the ability to control where and when a cell moves, using a simple beam of light, and control what it does when it gets there."
Many cell processes are governed by where and when proteins appear in the cell, Lim explained. When those processes are based on an extremely complex network of signals, such as in diseases like cancer, he added, it's helpful to have an on-off switch to insert into that process.
The research was carried out by Anselm Levskaya, a graduate student in both Lim's laboratory and in the laboratory of Chris Voigt, PhD, a synthetic biologist and assistant professor of pharmaceutical chemistry in the UCSF School of Pharmacy who was also a senior author on the paper.
Levskaya initially looked to plants for proteins that might serve as the light sensor. Plants are known to rely upon phytochromes, or light-sensing signaling proteins, to control a variety of processes, such as a plant's growth toward sunlight and seed germination.
He proposed that these phytochromes could be genetically engineered into mammalian cells and tied to a specific function, in this case, cell movement.
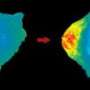
Levskaya identified a pair of interacting proteins from plants, known as the PhyB-PIF interaction, that could be turned on and off like a switch, and then imported that cellular signaling system into live mouse cells in a cellular pathway that controls cell motion. The resulting cells can be pulled by an external beam of dilute red light, or pushed away by an external infrared beam.
"We've been able to use similar light sensors to program bacteria and yeast cells to follow a chain of if-then commands," Voigt said. "What's remarkable here is the ability to, first, do this in mammalian cells, and secondly, find a method to turn them off again after they've performed the function we selected."
The reversible aspect of Levskaya's work is significant, Voigt said. While many methods are aimed at disrupting cellular pathways, most are fairly simple and only work in one direction: they shut a process down, or prevent two proteins from interacting, but they are limited to that one action.
This approach, by contrast, enables researchers to control precisely when the disruption occurs and for how long, then stop it at will.
The work involved a collaboration between three UCSF laboratories: the Voigt Laboratory, which uses synthetic biology to create light switches and sensors in bacteria; the Lim Lab, which studies how complex networks of signaling proteins cause cells to move, grow and differentiate; and the Weiner Lab, led by Orion Weiner, PhD, which uses microscopy to study guided cell movement.
Source: University of California - San Francisco