An impossible alloy now possible
What has been impossible has now been shown to be possible - an alloy between two incompatible elements. The findings are being published in this week's edition of Proceedings of the National Academy of Science, USA.
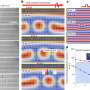
A research team led by Professor H.K. Mao from Carnegie Institution of Washington and Professor Rajeev Ahuja from UU have used high pressure experiments and theoretical calculations to study the behaviour of Ce3Al under high pressure.
"We were surprised to find that Cerium and Aluminium formed a so called substitutional alloy under high pressure. Forming these alloys has been limited to elements close in atomic radii and electronegativity up until now", sais Professor Rajeev Ahuja of Uppsala University.
The difference in radii and electronegativity of Cerium and Aluminium was diminished by applying pressure. Both synchrotron X-ray diffraction and ab initio calculations showed the same cause for bringing the two elements closer in radii and electronegativity, resulting in the new alloy phase. After the release of pressure the substitutional alloy still remained.
"This discovery opens up the possibility for finding new alloys with other ratios between Cerium and Aluminium, as well as alloys with Cerium and other incompatible elements. These new alloys may possess interesting and useful mechanical, electronic, and magnetic properties".
The new findings have been made possible by the recent development of high pressure diamond anvil cell and synchrotron radiation techniques. The extensive simulations were performed at Uppsala University's Multidisciplinary Center for Advanced Computational Science (UPPMAX).
More information: Read the article on PNAS website.
Source: Uppsala University