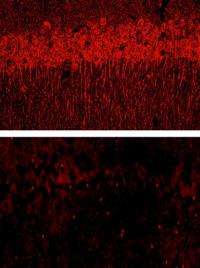
The extracellular matrix thins in mice lacking laminin in the hippocampus (bottom). Credit: Chen, Z.-L., et al. 2008. J. Cell Biol.
Shredded extracellular matrix (ECM) is toxic to neurons. Chen et al. reveal a new mechanism for how ECM demolition causes brain damage. The study will appear in the December 29, 2008 issue of The Journal of Cell Biology.
A stroke or head injury kills large numbers of neurons through a process called excitotoxicity. A surge of the neurotransmitter glutamate jolts receptors such as the kainate receptor and stimulates cell death. Enzymes add to the death toll by chopping up ECM near the injury site. How ECM breakdown takes out neurons was mysterious. The standard view was that neurons perished because they got separated from the ECM as it dissolved.
Chen et al. found otherwise when they engineered mice to lack the ECM component laminin in the hippocampus, a brain region often damaged by stroke or injury. If cells languished after parting from the ECM, the researchers reasoned that mice missing laminin would suffer more damage from excitotoxicity. But when excitotoxicity was spurred with an injection of kainate—a molecule that, like glutamate, activates the kainate receptor—the laminin-lacking mice showed less brain damage. After a dose of diced laminin, however, the mutant mice were vulnerable to kainate, indicating that the fragments are the culprit in cell death.
The researchers discovered that chopped-up ECM kills cells by ramping up production of one subunit of the kainate receptor, known as KA1. They speculate that hiking the amount of KA1 subunits might make the receptor more sensitive and thus more likely to trigger an overreaction by the cell.
Although drugs that obstruct the glutamate receptor slow brain cell death, they can lead to serious cognitive impairment and even coma. The study suggests that drugs that block KA1 might provide an alternative way to save brain cells after stroke or head trauma.
Paper: Chen, Z.-L., et al. 2008. J. Cell Biol. doi:10.1083/jcb.200803107, www.jcb.org
Source: Rockefeller University