Tiny buckyballs squeeze hydrogen like giant Jupiter
Hydrogen could be a clean, abundant energy source, but it's difficult to store in bulk. In new research, materials scientists at Rice University have made the surprising discovery that tiny carbon capsules called buckyballs are so strong they can hold volumes of hydrogen nearly as dense as those at the center of Jupiter.
The research appears on the March 2008 cover of the American Chemical Society's journal Nano Letters.
"Based on our calculations, it appears that some buckyballs are capable of holding volumes of hydrogen so dense as to be almost metallic," said lead researcher Boris Yakobson, professor of mechanical engineering and materials science at Rice. "It appears they can hold about 8 percent of their weight in hydrogen at room temperature, which is considerably better than the federal target of 6 percent."
The Department of Energy has devoted more than $1 billion to developing technologies for hydrogen-powered automobiles, including technologies to cost-effectively store hydrogen for use in cars. Hydrogen is the lightest element in the universe, and it is very difficult to store in bulk. For hydrogen cars to be competitive with gasoline-powered cars, they need a comparable range and a reasonably compact fuel system. It's estimated that a hydrogen-powered car with a suitable range will require a storage system with densities greater than those found in pure, liquid hydrogen.
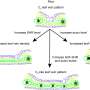
Yakobson said scientists have long argued the merits of storing hydrogen in tiny, molecular containers like buckyballs, and experiments have shown that it's possible to store small volumes of hydrogen inside buckyballs. The new research by Yakobson and former postdoctoral researchers Olga Pupysheva and Amir Farajian offers the first method of precisely calculating how much hydrogen a buckyball can hold before breaking.
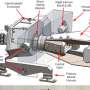
Buckyballs, which were discovered at Rice more than 20 years ago, are part of a family of carbon molecules called fullerenes. The family includes carbon nanotubes, the typical 60-atom buckyball and larger buckyballs composed of 2,000 or more atoms.
"Bonds between carbon atoms are among the strongest chemical bonds in nature," Yakobson said. "These bonds are what make diamond the hardest known substance, and our research showed that it takes an enormous amount of internal pressure to deform and break the carbon-carbon bonds in a fullerene."
Using a computer model, Yakobson's research team has tracked the strength of each atomic bond in a buckyball and simulated what happened to the bonds as more hydrogen atoms were packed inside. Yakobson said the model promises to be particularly useful because it is scalable, that is it can calculate exactly how much hydrogen a buckyball of any given size can hold, and it can also tell scientists how overstuffed buckyballs burst open and release their cargo.
If a feasible way to produce hydrogen-filled buckyballs is developed, Yakobson said, it might be possible to store them as a powder.
"They will likely assemble into weak molecular crystals or form a thin powder," he said. "They might find use in their whole form or be punctured under certain conditions to release pure hydrogen for fuel cells or other types of engines."
Source: Rice University