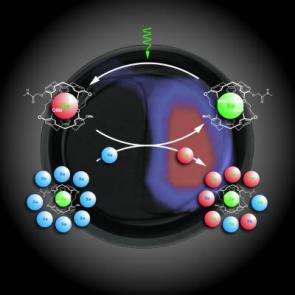
HYPER-CEST is a technique for enhancing the signal of molecular MRI through the combination of hyperpoalized xenon and the CEST technlogy. Credit: Lawrence Berkeley National Laboratory
Researchers with the U.S. Department of Energy's Lawrence Berkeley National Laboratory and the University of California at Berkeley have developed a new technique for Magnetic Resonance Imaging (MRI) that allows detection of signals from molecules present at 10,000 times lower concentrations than conventional MRI techniques.
Called HYPER-CEST, for hyperpolarized xenon chemical exchange saturation transfer, this new technique holds great promise for molecular imaging, in which the spatial distribution of specific molecules is detected within an organism. Ultimately, HYPER-CEST could become a valuable tool for medical diagnosis, including the early detection of cancer.
In a paper published in the October 20, 2006 issue of the journal Science, the team of researchers report on a technique in which xenon atoms that have been hyperpolarized with laser light to enhance their MRI signal, incorporated into a biosensor and linked to specific protein or ligand targets. These hyperpolarized xenon biosensors generate highly selective contrast at sites where they are bound, dramatically boosting the strength of the MRI signal and resulting in spatial images of the chosen molecular or cellular target.
This research was led by Alexander Pines and David Wemmer, who both hold joint appointments with Berkeley Lab and UC Berkeley. Their paper is entitled Molecular Imaging Using a Targeted Magnetic Resonance Hyperpolarized Biosensor. Co-authoring the paper with Pines and Wemmer were Leif Schröder and Thomas Lowery, plus Christian Hilty.
"Our HYPER-CEST molecular MRI technique makes optimum use of hyperpolarized xenon signals by creating a strong signal in regions where the biosensor is present, allowing for easy non-invasive determination of the target molecule," said Pines, one of the world's leading authorities on NMR/MRI technology, who holds a joint appointment as a chemist with Berkeley Lab's Materials Sciences Division and with UC Berkeley, where he is the Glenn T. Seaborg Professor of Chemistry. "This approach should be broadly applicable, potentially overcoming many shortcomings of currently used strategies for molecular imaging."
Added Wemmer, a chemist with Berkeley Lab's Physical Biosciences Division and UC Berkeley chemistry professor, "Other molecular MRI contrast agents provide small changes in big MRI signals, making the changes difficult to detect when the amount of contrast agent binding is small. Our HYPER-CEST contrast agent provides a big change in the xenon MRI signal, which means it is much easier to detect even though the xenon MRI signals are rather small."
In addition to its intrinsically higher contrast, another advantage with the HYPER-CEST technique is that its effects can be "multiplexed," meaning that the polarized xenon biosensors can be targeted to detect different proteins at the same time in a single sample. This capability, which is not shared by most conventional molecular MRI contrast agents, opens up a number of possibilities for future diagnostics.
Explained co-author Schröder, a member of the Pines' research group who is affiliated with Berkeley Lab's terials Sciences Division, "For example, as a diagnostic tool for the detection of cancer, with HYPER-CEST, we could perform multiple virtual biopsies on a single tissue sample, using different biosensors to screen for each potential form of cancer."
As a diagnostic tool for cancer, HYPER-CEST would be extremely sensitive, Schröder says, able to detect the presence of cancer-related proteins at micromolar (parts per million) concentrations. The sooner that the presence of cancerous cells is detected, the better the chances are for successful treatment. In addition to high sensitivity and target specificity, HYPER-CEST MRI is also unique from other molecular imaging techniques in that it provides both spatial and biochemical information. This points to a wide range of biomedical applications far beyond cancer diagnostics.
Said co-author Lowery, a member of the Wemmer research group, "We've demonstrated that HYPER-CEST is in the sensitivity ballpark for in vivo molecular imaging. In vivo molecular imaging is a very important tool if the dream of personalized medicine, where treatments are tailored not just to symptoms, but to the biochemical profile of an individual's disease state, is to be realized. By providing spatial and biochemical information, in vivo molecular imaging offers what other imaging technologies cannot, and this is where our HYPER-CEST MRI technique should really contribute."
MRI is well-established as a powerful technology for biomedical imaging. It is a painless and radiation-free means of obtaining high quality three-dimensional tomographical images of internal tissue and organs, particularly useful for opaque samples. However, the application of MRI to molecular imaging has been limited by sensitivity issues.
MRI, like its sister technology, nuclear magnetic resonance (NMR) spectroscopy, is based on a property of atomic nuclei with an unpaired proton or neutron called "spin." Such nuclei spin on an axis like miniature tops, giving rise to a magnetic moment, which means the nuclei act as if they were bar magnets with a north and south pole. When exposed to an external magnetic field, these spinning "bar magnets" attempt to align their axes along the lines of magnetic force. Since the alignment is not exact, the result is a wobbling rotation, or "precession," that's unique to each type of atom.
If, while exposed to the magnetic field, the precessing nuclei are also hit with a radiofrequency (rf) pulse, they will absorb and re-emit energy at specific frequencies according to their rate of precession. When the rf pulse is combined with magnetic field gradients a spatially encoded signal is produced that can be detected and translated into images.
Obtaining a spatially encoded MRI signal from a sample depends upon the spins of its precessing nuclei being polarized so that an excess point in one direction, either "up" or "down." Because the natural excess of up versus down spins for any typical population of atomic nuclei is only about one in 100,000, conventional MRI techniques are designed to detect nuclei that are highly abundant in tissue, usually the protons in water. Clinicians use contrasting agents to induce detectable changes in the MRI signal from a sample that can reveal the presence of anomalies. However, even though some of these contrasting agents will bind to specific biomolecular targets, the sensitivity is usually too low for molecular imaging.
In earlier studies, Pines and his group exploited the fact that zapping rubidium vapor with a beam of polarized laser light creates a "hyperpolarized" effect that can be transferred to nuclei of xenon, an inert gas whose nuclei naturally feature a tiny degree of spin polarization. This process, called "optical-pumping," vastly increases the proportion of spin-up nuclei, producing a population of xenon atoms with nearly 50-percent of their nuclei in the up state. Pines and his group also developed techniques for transferring this hyperpolarization from xenon nuclei to other molecules, and methods to probe the environments of the xenon atoms and their movement.
Working with the Pines group, Wemmer and his group subsequently used a hollow molecular cage, called a cryptophane, which could be specially adapted to hold hyperpolarized xenon atoms. With the addition of a biochemical "linker" that makes the cage soluble in water, they created a novel agent that binds to a specific target molecule and associates the hyperpolarized xenon with it. These sensitive and versatile xenon biosensors can then be used to selectively alter MRI signals.
In the new Science paper, the Pines and Wemmer collaboration combined the xenon biosensors with CEST technology, which was developed as a contrast method for boosting the MRI contrast from protons. CEST is based on the exchange of protons that takes place between water molecules and other molecules in the body. (Approximately 60-percent of the human body and 90-percent of the blood is water.) During an MRI session, these exchanging protons can be selectively labeled with the RF pulse. Comparing MRI images taken with and without CEST labeling yields images with meaningful bright contrasts. While effective under certain conditions, CEST is limited by the fast relaxation time of protons, which necessitates the need for large magnetic fields to decrease relaxation effects and increase the difference between saturated and non-saturated MRI signals. It also requires a relatively large amount of the CEST agent to generate significant contrast enhancement.
Hyperpolarized xenon has a much longer relaxation time than protons, which means that the enhanced MRI signal is not only stronger, but lasts much longer. The MRI signal obtained directly from the xenon biosensors is hundreds of times smaller than the easily detected signal obtained from a pool of free xenon dissolved in the rest of the sample. The HYPER-CEST images are based on the free xenon signal rather than direct detection of the biosensors which leads to the high sensitivity of the technique.
Another huge advantage, as Lowery explained, is that "there is no xenon naturally present in body, so we don't have to fish out a small change in an MRI signal with a high background like other MRI contrast agents. In that sense, HYPER-CEST is more like a PET (Positron Emission Tomography) probe. Because multiple xenon biosensors can be used to detect different targets at the same time, HYPER-CEST is also like quantum dots, where different colors of dots can be used to simultaneously report different analytes. However, unlike quantum dots, our xenon agents can be detected deep within the body."
The Berkeley HYPER-CEST technique not only provides a bright image with high contrast, but also speeds up the image acquisition process by better than three-thousand fold relative to directly detecting the xenon biosensor.
Said Schröder, "Image acquisition time becomes a matter of minutes as opposed to a matter of days under conventional molecular MRI techniques. Our HYPER-CEST technique is the first rapid means of imaging the spatial distribution of a target molecule, and should lend itself to advanced clinical diagnostics. For example, we could target a molecule that sits on the surfaces of cancer cells to obtain critical morphological information. Improving the biosensor construct, it may even be possible to eventually bring our sensitivity down to a single cell."
For their Science paper, the Berkeley researchers demonstrated their HYPER-CEST technique on a two-compartment phantom containing avidin-agarose beads. The next step is to perform a version of this experiment on biological cells in culture. Lowery is already in the process of developing antibody-targeted probes for this stage of the research.
"Once we've completed the experiments on cells in culture, we will look to do xenon biosensor molecular imaging in an animal model, probably mice," said Lowery. "Based on previous work done by the Pines group, we know how to deliver the hyperpolarized xenon in vivo, either by injection or inhalation. All we need to do is to introduce a targeted sensor to our animal model before we do the MRI imaging. After the first in vivo xenon biosensor images have been demonstrated, then development can become serious for diagnostic applications."
Said Wemmer, "For in vivo applications HYPER-CEST can provide MRI type signals associated with specific ll types of any kind that have known distinctive surface proteins markers. Our results mark a critical step toward the application of xenon biosensors as selective molecular MRI contrast agents for biomedical applications."
Source: Lawrence Berkeley National Laboratory