Quantum Dots Pose Minimal Impact to Cells

Nano-sized fluorescent probes that can slip inside living cells and elucidate life’s most fundamental processes, or track the effectiveness of cancer-fighting drugs, are barely noticed by the cells they enter, according to a team of researchers led by the U.S. Department of Energy’s Lawrence Berkeley National Laboratory.
Using a high-throughput gene expression test, the team determined that the probes, which are specially coated quantum dots, only affect 0.2 percent of the human genome. This finding should quell concerns that the mere presence of these promising but potentially toxic sentinels disrupts a cell’s function, confounding quantum dots’ ability to accurately track cellular processes or monitor the effectiveness of pharmaceuticals.
“Because of their protective coating, we found that quantum dots pose minimal impact to cells,” says Fanqing Frank Chen, a scientist in Berkeley Lab’s Life Sciences Division who led the research team. “The only gene changes we see are related to transporting the dots into and within cells.”
In addition, the tool used by Chen and colleagues to analyze quantum dots — a gene chip packed with 18,400 probes of known human genes — is one of the world’s most comprehensive and streamlined ways to measure the toxicity of nano-scale particles. This is an especially important tool given that nanoparticles frequently make the news over concerns that they pose health risks.
“Berkeley Lab is one of the first labs in the world to conduct and publish studies on high-throughput, whole-genome analyses of the toxicity of nanoparticles” says Chen.
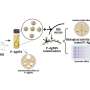
Chen’s team used this toxicogenomics tool to study quantum dots, which are crystalline semiconductors composed of a few hundred or thousand atoms that emit different colors of light when illuminated by a laser. Because these fluorescent probes are stable, they have the ability to remain in a cell’s cytoplasm and nucleus without fading out much longer than conventional fluorescent labels. This could give biologists a clear view of processes that span several hours or even days, such as DNA replication, genomic alterations, and cell cycle control. Their longevity has also made quantum dots a powerful molecular label, allowing scientists to study the earliest signs of cancer, track the effectiveness of pharmaceuticals that target the cellular underpinnings of disease, and understand the events that occur during stem cell differentiation.
Several years ago, Paul Alivisatos, a Berkeley Lab chemist in the Materials Sciences Division and Associate Laboratory Director, developed a way to fashion especially stable quantum dots from cadmium selenide and zinc sulfide. One drawback to this approach, however, is that these quantum dots may release potentially toxic cadmium and zinc ions into cells.
To solve this problem, Alivisatos and Daniele Gerion, a former postdoc in the Alivisatos lab, coated the dots with a protective layer of polyethylene glycol, which is a very nonreactive and stable compound that is used extensively by the pharmaceutical industry in drug formulation. This layer is designed to prevent the dots from leaking heavy metal ions into cells once they’re inside.
“The polyethylene glycose compound does not break down easily. At a very small scale, it is almost perfect in structure,” says Chen.
To test how well this coating does its job, Chen’s team recently turned to the tiny chip filled with several thousands probes of known human genes. This chip enables the researchers to quickly expose the human genome to a compound, such as quantum dots, and determine the extent to which the compound forces the genes to express themselves abnormally.
Their work is part of a new field called toxicogenomics. It’s based on the idea that if the environment inside a cell is altered by an external stimulus, then some of the cell’s genes will likely express themselves in an atypical way. The more toxic the external stimulus, the greater the number of genes that will be altered. Conversely, if the stimulus is benign, then very few genes will change. With this in mind, Chen’s team introduced polyethylene glycose-coated dots inside living cells, and ran the gene expression test.
“We found that of the 18,400 genes on our chip, only approximately 50 genes were affected, which is about 0.2 percent of the human genome,” says Chen.
According to Chen, this miniscule shift isn’t worrisome for several reasons. First, the number of genes affected is very small given the large dose of quantum dots used in the study, which is up to 1000 times greater than the dose that would typically be used. Second, the affected genes are not related to heavy metal exposure, which would be the case if the cells had been exposed to cadmium or zinc ions. And third, the genes that do change are involved in transporting the quantum dots through the cell membrane and within the cell.
“We see changes in transporter proteins, which is expected because the dots have to be transported into and within the cell,” says Chen, who reported this research in an April issue of the journal Nano Letters. Based on their results, Chen’s team hopes to soon use quantum dots for in vivo imaging of breast and prostate cancer.
“We could use quantum dots to see cancers at very early stages, as well as characterize the molecular makeup of cancer,” says Chen.
The Berkeley Lab team is also working in collaboration with other researchers on the mutagenic and carcinogenic characterization of these silica-coated quantum dots.
Researchers from the Santa Clara, California-based company Affymetrix, Lawrence Livermore National Laboratory, the University of California at Berkeley, and the University of California at San Francisco’s Comprehensive Cancer Center also participated in the study.
Source: Berkeley Lab