Chemical Could Revolutionize Polymer Fuel Cells
Heat has always been a problem for fuel cells. There’s usually either too much (ceramic fuel cells) for certain portable uses, such as automobiles or electronics, or too little (polymer fuel cells) to be efficient.
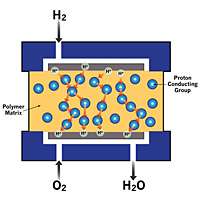
Image: Ddiagram of a fuel cell’s polymer electrolyte membrane (PEM) with the proton-conducting group triazole (the circles in the diagram). Protons hop from one group to another to move through the PEM without the need of water.
While polymer electrolyte membrane (PEM) fuel cells are widely considered the most promising fuel cells for portable use, their low operating temperature and consequent low efficiency have blocked their jump from promising technology to practical technology.
But researchers at the Georgia Institute of Technology have pinpointed a chemical that could allow PEM fuel cells to operate at a much higher temperature without moisture, potentially meaning that polymer fuel cells could be made much more cheaply than ever before and finally run at temperatures high enough to make them practical for use in cars and small electronics.
A team lead by Dr. Meilin Liu, a professor in the School of Materials Science and Engineering at Georgia Tech, has discovered that a chemical called triazole is significantly more effective than similar chemicals researchers have explored to increase conductivity and reduce moisture dependence in polymer membranes. The findings were published in the Journal of the American Chemical Society.
“Triazole will greatly reduce many of the problems that have prevented polymer fuel cells from making their way into things like cars, cell phones and laptops,” said Liu. “It’s going to have a dramatic effect.”
A fuel cell essentially produces electricity by converting the chemicals hydrogen and oxygen into water. To do this, the fuel cell needs a proton exchange membrane, a specially treated material that looks a lot like plastic wrap, to conduct protons (positively charged ions) but block electrons. This membrane is the key to building a better fuel cell.
Current PEMs used in fuel cells have several problems that prevent them from wide use. First, their operating temperature is so low that even trace amounts of carbon monoxide in hydrogen fuel will poison the fuel cell’s platinum catalyst. To avoid this contamination, the hydrogen fuel must go through a very expensive purification process that makes fuel cells a pricey alternative to conventional batteries or gasoline-fueled engines. At higher temperatures, like those allowed by a membrane containing triazole, the fuel cell can tolerate much higher levels of carbon monoxide in the hydrogen fuel.
The use of triazole also solves one of the most persistent problems of fuel cells — heat. Ceramic fuel cells currently on the market run at a very high temperature (about 800 degrees Celsius) and are too hot for most portable applications such as small electronics.
While existing PEM fuel cells can operate at much lower temperatures, they are much less efficient than ceramic fuel cells. Polymer fuel cell membranes must be kept relatively cool so that membranes can retain the moisture they need to conduct protons. To do this, polymer fuel cells were previously forced to operate at temperatures below 100 degrees Celsius.
Heat must be removed from the fuel cells to keep them cool, and a water balance has to be maintained to ensure the required hydration of the PEMs. This increases the complexity of the fuel cell system and significantly reduces its overall efficiency. But by using triazole-containing PEMs, Liu’s team has been able to increase their PEM fuel cell operating temperatures to above 120 degrees Celsius, eliminating the need for a water management system and dramatically simplifying the cooling system.
“We’re using the triazole to replace water,” Liu said. “By doing so, we can bring up the temperature significantly.”
Triazole is also a very stable chemical and fosters stable fuel cell operating conditions.
While they have pushed their polymer fuel cells to 120 degrees Celsius with triazole, Liu’s team is looking into better polymers to get those temperatures even higher, he said.
Source: Georgia Institute of Technology