Light oscillations become visible
The human eye can detect changes in the intensity of light, not however the wavelength because light oscillates too fast (approximately 1000 trillion times per second). An international collaboration led by Ferenc Krausz and made up of researchers from the Vienna University of Technology, the Max-Planck-Institute for Quantum Optics and the University of Bielefeld have recently succeeded in developing a technique which can measure the instantaneous electric field of red light (quarter period ~ 620 attoseconds) and record its variation with a resolution of 100 attoseconds (Science, August 27, 2004). The experiment of the Austrian-German team allowed the first direct visualization of the electric field of visible light and constitutes the fastest measurement to date.
It has been known since the famous experiments of Heinrich Hertz near the end of the 19th century that light is a wave consisting of electric and magnetic fields, just as radio waves and microwaves. The only difference is in the number of times these fields change their direction in a second. In radio and microwaves this happens typically millions to trillions times per second. The field variation in these waves can be readily detected by turning it into electric current and displaying the variation of this current in electronic instruments called oscilloscopes.
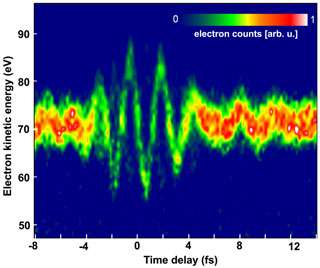
Fig. 1: Energy shift (in units of eV) suffered by an attosecond electron probe set free at different instants (measured in units of fs) in an intense wave consisting of only a few cycles of red light.
In striking contrast, the electromagnetic field of visible light changes direction approx. one thousand trillion, i.e. 1 000 000 000 000 000, times per second, so that the instantaneous intensity of the light field varies from zero to maximum faster than a femtosecond (1 femtosecond being one thousandth of a trillionth of a second), some ten thousand times more rapidly than the resolution of the fastest electronic instruments available to date. Recording the field variation of visible light calls for an oscilloscope that exhibits a temporal resolution of several hundred attoseconds (1 attosecond being a thousandth of a femtosecond). The researchers recently succeeded in developing a technique which can measure the instantaneous electric field of red light (quarter period ~ 620 attoseconds) and record its variation with a resolution of 100 attoseconds.
The key to this measurement was the generation of single 250-attosecond extreme ultraviolet pulses, a feat achieved by the same collaboration a few months ago (Nature, February 26, 2004). The attosecond extreme ultraviolet pulse knocks electrons free from atoms to probe the electric field of a wave consisting of only a few cycles of red laser light. The electric field of red light accelerated or decelerated the electrons set free with respect to the light wave with a 100-attosecond timing precision. The change in the electrons’ energy (shown in units of electron volts, eV, in Fig. 1), measured as a function of delay (shown in units of femtoseconds, fs, in Fig. 1) between the attosecond pulse and the laser light wave clearly exhibits the build-up and disappearance of the laser pulse within a few femtoseconds as well as oscillations with a period of the 2.5-fs wave cycle of 750-nm (red) light. The measured energy change directly yields the variation of the instantaneous strength and direction of the electric field of the few-cycle light wave (Fig. 2).
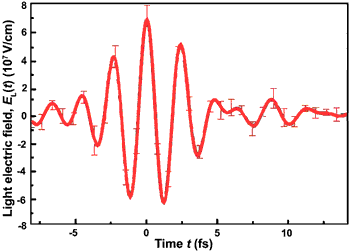
Fig. 2: Build-up and disappearance of the electric field in the 4.3-fs pulse of red light (wavelength ~ 750nm), as recorded by the attosecond oscilloscope.
The red line in Fig. 2 depicts the electric field of a few-femtosecond flash of red light, as recorded by an apparatus that can be regarded as the first attosecond oscilloscope. The new technique permits direct and accurate measurement of ultrabroad-band light pulses (made up of many different colours), and thereby opens the door to the reproducible synthesis of ultrashort flashes of light with arbitrary waveform for a number of applications including the development of molecular electronics and X-ray lasers.
Source: Max-Planck-Institute of Quantum Optics















