Pulsing light silences overactive neurons

Scientists at the MIT Media Lab have invented a way to reversibly silence brain cells using pulses of yellow light, offering the prospect of controlling the haywire neuron activity that occurs in diseases such as epilepsy and Parkinson's disease.
Such diseases often must be treated by removing neurons that fire incorrectly. The new MIT research could lead to the development of optical brain prosthetics to control neurons, eliminating the need for irreversible surgery.
"In the future, controlling the activity patterns of neurons may enable very specific treatments for neurological and psychiatric diseases, with few or no side effects," said Edward Boyden, assistant professor in the Program in Media Arts and Sciences and leader of the Media Lab's new Neuroengineering and Neuromedia Group.
Boyden and Media Lab research affiliate Xue Han published their results in the March 21 issue of the online journal Public Library of Science ONE.
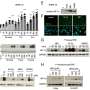
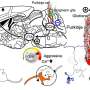
The work takes advantage of a gene called halorhodopsin found in a bacterium that grows in extremely salty water, such as the Great Salt Lake in Utah. In the bacterium, Natronomas pharaonis, the gene codes for a protein that serves as a light-activated chloride pump, which helps the bacterium make energy.
When neurons are engineered to express the halorhodopsin gene, the researchers can inhibit their activity by shining yellow light on them. Light activates the chloride pumps, which drive chloride ions into the neurons, lowering their voltage and silencing their firing.
That inhibitory effect may be extremely useful in dealing with diseases caused by out-of-control neuron firing, said Boyden. "In such diseases, inhibition is more direct than excitation, because you can shut down neural circuits that are behaving erratically," he said.
Many epilepsy patients have implanted electrodes that periodically give their brains an electric jolt, acting as a defibrillator to shut down overactive neurons. This new research opens up the possibility of an optical implant that could do the same thing, using light instead of electricity. The Media Lab neuroengineering group plans to start studying such devices in transgenic mice this year.
The group also plans to use the new method to study neural circuits. Last year, Boyden devised a technique to stimulate neurons by shining blue light on them, so with blue and yellow light the researchers can now exert exquisite control over the stimulation and inhibition of individual neurons.
Learning more about the neural circuits involved in epilepsy could help scientists develop devices that can predict when a seizure is about to occur, allowing treatment (either shock or light) to be administered only when necessary, Boyden said.
The technique also offers a way to study other brain diseases, as well as normal brain circuitry, offering insight into which brain regions and neurons contribute to specific behaviors or pathological states, Boyden said.
The halorhodopsin gene was originally discovered in the 1980s, but Boyden didn't think its full potential had been explored. The protein expressed by the gene turned out to have exactly the right characteristics to make it useful in neuron inhibition.
"Often if you are patient and think carefully about what you want to do, you can find a molecule that is very close to what you want, and with a little bit of luck it will turn out to work," Boyden said.
The halorhodopsin work is one of the first projects from the Media Lab Neuroengineering and Neuromedia Group, which was formed about six months ago to enhance the Media Lab's study of the brain-body relationship.
"The Media Lab has always been interested in studying the interface between people and the world," Boyden said, "but now people are getting interested in the interface between bodies and brains in the world."
The research was funded by an anonymous donor, the MIT Media Lab and the Helen Hay Whitney Foundation.
Source: MIT