Scientists discover genes that can slow cell division and may fight cancer
Cancer cells differ from normal cells in, among other things, the way they divide. When a normal cell complies with a signal telling it to divide, it also begins to activate a "braking system" that eventually stops cell division and returns the cell to a resting state. When that braking system is faulty, uncontrolled cell division and the growth of cancer can result. Weizmann Institute scientists studied this system of brakes, and identified a number of the genes involved.
According to the study's findings, which appeared in Nature Genetics online on February 25, aberrations in the activities of these genes are tied to certain types of cancer, as well as to the relative aggressiveness of the cancer. These insights may, in the future, lead to the development of ways to restore the brakes on runaway cell division and halt the progression of cancer.
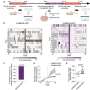
First, the scientists mapped the network of genes that is activated in normal cells upon receiving the order to divide. The "divide!" signal comes from outside the cell in the form of a chemical called a growth factor, and it initiates a chain of events inside the cell. The genes activated in this sequence produce proteins, some of which cause cell division and others that put the brakes on that division. To find which genes were responsible, the scientists needed to sift through a huge quantity of data on genes and their activities. To cope with this monumental task, a team of Weizmann Institute researchers from diverse fields pooled their knowledge and experience: Prof. Yosef Yarden of the Biological Regulation Department, Prof. Eytan Domany of the Physics of Complex Systems Department, Prof. Uri Alon of the Molecular Cell Biology Department, and Dr. Eran Segal of the Computer Science and Applied Mathematics Department. Working with them were Prof. Gideon Rechavi of the Sheba Medical Center and researchers from the M.D. Anderson Cancer Center in Houston, Texas, headed by Prof. Gordon B. Mills.
This collaboration between physicists, mathematicians, computer scientists, and biologists – the sort of multidisciplinary research for which the Weizmann Institute has gained a global reputation – yielded some startling results. They found that following the receipt of the growth factor signal, cell activity takes place in a number of separate waves in which genes are turned on and off for different periods of time. In the first wave, the activity of a few genes rises for about 20 to 40 minutes. These are the genes that cause the cell to divide. In contrast, the next four waves, ranging from 40 to 240 minutes after the signal, are comprised primarily of gene activity tied to the process of halting cell division.
The scientists then focused on identifying the genes in these later waves and confirming that they do, indeed, put the brakes on cell division. Through their wide-ranging study, they found 50 genes that interfere with the genetic activities of the first wave. This braking system works by producing proteins that directly attach to the cell-division genes, hindering their activity. Yet another protein they identified works, instead, by dismantling messenger RNA carrying instructions for making cell division proteins from the genes to the cell's protein-production machinery.
In tests conducted on tissues from ovarian cancer patients, the scientists found a correlation between levels of activity in the "braking" genes, rates of survival, and the aggressiveness of the disease. These findings point the way toward the development of a personal genetic profile that might pinpoint the genetic defects responsible for each cancer and help doctors tailor a treatment fitted to each patient. Such a genetic profile can also help predict the individual progression of the disease. In the future, the identification of the exact factors causing uncontrolled cell division in different cancers might lead to the development of effective treatments for preventing or halting cancer growth.
Source: American Committee for the Weizmann Institute of Science