Nanoscale packaging could aid delivery of cancer-fighting drugs
Nature has produced a well-stocked arsenal of potent cancer-fighting compounds, including Taxol, first isolated from the Pacific yew tree, and rapamycin, borrowed from a soil-dwelling bacterium.
But there's a catch. Many natural anti-cancer molecules are large, complex and fat-soluble, making them tough to administer to patients and keep circulating in the blood long enough to attack their targets.
Working in the emerging field of "nanomedicine," University of Wisconsin-Madison pharmacy professor Glen Kwon aims to improve the delivery of drugs like these by targeting them more selectively to tumors and boosting their solubility in water. Standard formulations exist but involve dissolving drugs in organic solvents like ethanol or with soap-like molecules called surfactants.
"Think about surfactant or soap being injected into a patient," says Kwon. "But we do that for a lot of anti-cancer drugs, although we are getting more sophisticated."
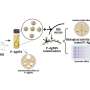
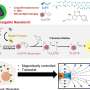
Kwon instead coaxes water-insoluble drugs inside nanoscale spheres, called polymeric micelles, which can circulate in the bloodstream for long periods. Like soap, the polymers composing his micelles contain a "hydrophobic" region that repels water and a "hydrophilic" region that attracts it. In water, the polymers spontaneously assemble into tiny spheres, each with a hydrophobic center and a hydrophilic outer shell.
Soap dissolves grease in water by bringing oil and fat inside the core of its micelles. Kwon's approach works the same way: When mixed with a fat-soluble drug, polymeric micelles convey the compound inside. Enveloped by the micelle's hydrated outer shell, the drug then becomes much more water-soluble than it would be normally.
Unlike soap, however, Kwon's micelles are composed of benign and biocompatible polymers, including a biodegradable compound that is already used for long-term release in birth control.
Besides being safer and easier to administer, polymeric micelles maintain anti-cancer drugs like rapamycin in blood plasma for longer periods than do standard formulations, Kwon has found. It's a promising result that could give the drugs a greater chance of accumulating at tumor sites.
Polymeric micelles may also make it easier to mix stronger cancer-fighting cocktails containing more than one chemotherapeutic agent. Doing so now is a challenge because hydrophobic drugs in solution together tend to "crash out," says Kwon, becoming particulate, aggregated — and useless.
"We've set the stage for concurrent, combination drug therapy," he says. "These are all drugs that are proven already, but from a delivery point of view we think we can make an improvement."
Source: by Madeline Fisher, University of Wisconsin-Madison