How blood flow dictates gene expression
Researchers at the University of Pennsylvania School of Medicine have pinpointed a key regulatory protein that translates blood flow into gene expression. The investigators showed that in a model of mouse embryonic development a transcription factor called Klf2, which resides in cells that line blood vessels, is activated by rapid, pulsed blood flow, as reported in the December issue of Developmental Cell. Understanding Klf2's role in blood vessel and muscle biology could help with fighting atherosclerosis.
"We always knew that there had to be this line of communication from the vessel lining, or endothelium, to the smooth muscles, which never sees a blood cell," says senior author Mark Kahn, MD, Associate Professor of Medicine. "That's where Klf2 fits: This is the first time, at a molecular level, that this chain has been demonstrated in an animal."
Swirling eddies of blood form when vessels branch, much like when a river divides. Atherosclerosis typically forms at these sites of so-called disturbed flow as opposed to regions of rapid blood flow through the main vessels. This relationship between atherosclerosis and flow has been known for decades. More recently, tissue- culture studies have shown that Klf2 is activated by increased blood flow, or "fluid sheer stress."
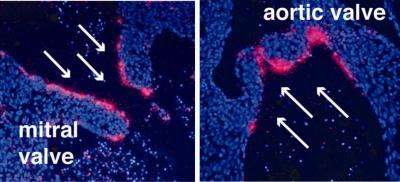
Indeed, in this study Kahn; first author John S. Lee, MD, PhD, Instructor in the Department of Medicine; and colleagues show that the expression of Klf2 in a developing mouse embryo mirrors events in previous tissue-culture studies. They found that Klf2 is expressed on the high-flow side of developing mitral and aortic valves in the heart of a 14-day-old embryo.
The researchers surmise that the mechanical stimulus of blood flowing in a vessel leads to the upregulation of Klf2, which either activates or represses genes that control smooth muscle tone, that is the caliber of the vessel. (Tone is governed by how much a muscle contracts or relaxes.) These genes encode proteins that are either secreted or are on the cell surface of the endothelium and so influence how smooth-muscle cells contract or relax.
The researchers suggest that when Klf2 is expressed, smooth muscle cells lining the blood vessels maintain their ability to regulate vessel tone. However, when Klf2 is genetically deleted, or "knocked out," from the blood vessels of mouse embryos, they had an abnormally high cardiac output, as measured by ultrasound, while the overall structure of their blood vessels was normal. These findings implicate loss of vessel tone as the primary defect in Klf2 knockout mice.
Now that Klf2 has been established as an important regulator of blood flow in live animals and is required for the development of a healthy cardiovascular system, the next step is to elucidate the role of Klf2 in normal adult blood vessels and in the pathogenesis of vascular diseases, such as atherosclerosis.
Source: University of Pennsylvania School of Medicine