Two Central Mysteries in Genome Inheritance Solved at UCSD
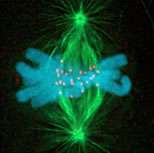
The dance of the chromosomes during cell division, first described in the late 1800s and familiar to all high-school students from movies shown in biology classes, has long fascinated biologists. However, the molecular nature of a key component of cell division, the “chromosome-spindle” connection, which is critical for the inheritance of genetic information as cells divide, has remained elusive.
Researchers at the University of California, San Diego (UCSD) School of Medicine and the Ludwig Institute for Cancer Research have identified a protein group that forms the chromosome-spindle connection involved in pulling apart the two replicas of the duplicated genome during cell division. They have also identified a separate protein complex that helps the cell discriminate between correct and incorrect spindle connections, and shuts down the pulling process when the connections are incorrect to ensure that each new cell has a complete set of chromosomes.
In a pair of papers in the journal Cell – one published in the December 1 issue; the second on December 15 – the scientists report these solutions to two long-standing mysteries about the mechanism of genome inheritance during cell division.
“These two discoveries open the door to further studies of the chromosome-spindle interface, studies that could result in improvement of chemotherapies for cancer,” said Arshad Desai, Ph.D., assistant professor of Cellular and Molecular Medicine at UCSD and investigator with the Ludwig Institute, who is principle investigator of both studies.
Cell division is central to the development, maintenance and propagation of all living organisms. During every cell cycle, the genome – which carries the blueprint of life – must be duplicated and distributed. Errors in genome distribution result in genetic birth defects and contribute to the genesis of cancer.
Accurate distribution of the genome, which is split into chromosomes of varying number depending on the species, requires specialized protein polymers called microtubules.
“After the genome is replicated, microtubule polymers build a spindle-shaped structure whose two ends collect precisely half of the duplicated genome before the cell is pinched in the middle to form two cells,” said Desai. Chromosomes connect to the spindle microtubule polymers during this time, and interfering with this connection – using drugs that bind to microtubule polymers – is a common chemotherapy strategy used to target and curtail rapidly dividing cancer cells. While scientists have known that breakdown of this connection could stop the proliferation of cancerous cells, they didn’t know how the connection itself was established.
Using a biochemical approach, Desai and postdoctoral fellow Iain Cheeseman, Ph.D., have solved this mystery. In the December 1 paper, they identified a protein group that forms the chromosome-spindle connection and is present in even the simplest single-celled organisms, indicating that its role in genome distribution is ancient and widely conserved. Identification of this protein group provides scientists with new drug targets in cancer cells.
“Right now, drugs used in chemotherapy affect microtubules in all the body’s cells, resulting in adverse side effects, like nerve pain and loss of sensation ,” said Desai. “If we could specifically target dividing cells, we should improve on current chemotherapy to treat the cancer with fewer side effects.”
The second paper from Desai’s research group, to be published in the December 15 issue of Cell, identifies a different type of connection between chromosomes and microtubule polymers, which operates to discriminate between correct and incorrect chromosome-spindle attachments.
Each replicated chromosome is comprised of a pair of identical sister chromatids, generated by the copying of the parental DNA strand. These pairs are held together during the early stages of cell division. Once all chromosomes properly connect to the spindle, the pairs are separated and sisters are pulled towards opposite ends of the spindle.
“If both chromatids of a chromosome, by chance, connect to a single end of the football-shaped spindle, the resulting daughter cells will lack the correct complement of genomic information – a situation that is dangerous as it can promote cancerous transformation,” said Desai.
Cells avoid such a fate by detecting errors in attachment to the spindle, breaking down these defective attachments and starting over. But until now, scientists did not understand how cells detected such an error.
Sharsti Sandall, a graduate student in the Biomedical Sciences Program at UCSD, identified a complex of two proteins that bridges chromosomes and spindle microtubules and is required to detect attachment errors. This complex controls activation of a protein kinase called Aurora.
“A major question becomes, how are bad attachments detected?” said Desai. “Our results suggest that the linkage between chromosomes and the spindle, which includes a protein kinase activator, acts a sensor for bad attachments and relays their presence into kinase activation.”
A protein kinase transfers a phosphate group to target proteins in order to modify their properties. One of the targets of activated Aurora kinase is the protein group that makes the chromosome-spindle connection, identified by Cheeseman in the December 1 paper.
Aurora phosphorylation reduces the ability of this protein group to bind microtubule polymers. This explains how bad attachments are dissolved, prompting new tries until all chromosomes in the cell are properly connected. Desai’s future goal is to discover precisely how the complex identified by Sandall activates Aurora kinase only near bad attachments.
Additional researchers include: (Cheeseman et al) Joshua S. Chappie and Elizabeth M. Wilson-Kubalek, Department of Cell Biology, The Scripps Research Institute (Shandall et al) Fedor Severin, Ph.D. and Anthony A. Hyman, Ph.D., Max Plank Institute; Ian X. McLeod, Ph.D., and John R. Yates, Ph.D., The Scripps Research Institute; and Karen Oegema, Ph.D., Ludwig Institute for Cancer Research and UCSD School of Medicine.
Source: UCSD, by Debra Kain





















