Alloy of hydrogen and oxygen made from water
Water, the only indispensable ingredient of life, is just about the most versatile stuff on Earth. Depending on its temperature we can heat our homes with it, bathe in it, and even strap on skates and glide across it, to name only the most common of its many forms. When subjected to high pressures, however, water can take any of more than 15 different forms.
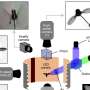
Researchers have now used x-rays to dissociate water at high pressure to form a solid mixture--an alloy--of molecular oxygen and molecular hydrogen. The work, by a multi-institutional team that includes Russell Hemley and Ho-kwang Mao of Carnegie's Geophysical Laboratory, appears in the October 27 issue of Science.
The researchers subjected a sample of water to extremely high pressures--about 170,000 times the pressure at sea level (17 Gigapascals)--using a diamond anvil, and zapped it with high-energy x-rays. Under these conditions, nearly all the water molecules split apart and re-formed into a solid alloy of O2 and H2. X-radiation proved to be the key to cleaving the O-H bonds in water; without it, the water remained in a high-pressure form of ice known as ice VII--one of at least 15 such variants of ice that exist under high pressure and variable temperature conditions.
"We managed to hit on just the right level of x-ray energy input," explained Hemley. "Any higher, and the radiation tends to pass right through the sample. Any lower, and the radiation is largely absorbed by the diamonds in our pressure apparatus."
This rather narrow range of energy requirement explains why, in hundreds of previous high-pressure x-ray experiments, the breakdown reaction had gone undiscovered: most such experiments tend to use more energetic x-rays. The experiments also required long, multiple-hour irradiation with x-rays; such long exposures had not been attempted before.
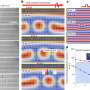
The researchers put the alloy through its paces, subjecting it to a range of pressure, temperature, and bombardment with x-ray and laser radiation. As long as the sample remained under pressure equivalent to about 10,000 times atmospheric pressure at sea level (1 Gigapascal), it stood up to this punishment. Although the substance is clearly a crystalline solid, more experiments are needed to determine its precise crystal structure.
"The new radiation chemistry at high pressure was surprising," said lead author Wendy Mao of Los Alamos National Laboratory. "The new alloy containing the incompatible oxygen and hydrogen molecules will be a highly energetic material."
Source: Carnegie Institution