Scientists unravel 'molecular inch-worm' structure of walking-pneumonia bacterium
Researchers at the University of Georgia, using glow-in-the-dark proteins and microcinematography, have helped unravel the development and function of a complex organelle in the bacterium that causes “walking pneumonia.”
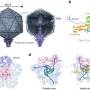
The researchers have described in new, precise detail the unique cell extension that forms on one end of the bacterium Mycoplasma pneumoniae. This structure, called a “terminal organelle,” performs several tasks for this pervasive bacterium and even acts as a “molecular inch-worm,” helping the microorganism move.
“Mycoplasmas are among the simplest known prokaryotes—only a fraction the size of other health-related bacteria such as E. coli,” said microbiologist Duncan Krause, leader of the research team. “They are true minimalists with very small genomes, lacking the typical cell regulatory mechanisms found in other bacteria. And yet some species such as M. pneumoniae posses this complex terminal organelle. We’ve been able to observe it in growing cultures and describe the choreography of events at a level of detail not previously possible.”
The research is being published this week in The Proceedings of the National Academy of Sciences. Other authors of the paper include graduate student Benjamin Hasselbring, undergraduate Robert Krause and former graduate student Jarrat Jordan.
M. pneumoniae infections affect millions worldwide, causing chronic bronchitis and atypical or “walking pneumonia,” a term that describes cases of pneumonia that are distinct from acute, life-threatening pneumonia requiring a patient’s hospitalization.
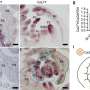
Krause and others have been increasingly interested in the terminal organelle that develops on one end of M. pneumoniae because it is involved in cell division, adherence to respiratory tissues and a little-understood mechanism of propulsion called “gliding motility.”
Bacteria can move in a variety of ways, including the use of flagella to “swim.” But since M. pneumoniae lack flagella, they “glide,” a method of movement that has been known for some time yet never entirely understood. The cells seem to bend and flex, but it’s unclear how that is accomplished. The new data indicate that gliding is essential for cell division in M. pneumoniae.
“In addition to its significant impact on public health, M. pneumoniae is intriguing from a biological perspective,” said Krause. “They have no cell walls, and their genome is among the smallest known for a cell capable of a free-living existence.”
Other researchers, using electron microscopy, have described the basic structure of the terminal organelle, but Krause’s team went further, using fluorescence microscopy and fluorescent protein fusions that allowed them to track the actions of specific proteins in live, growing cells. Time-lapse digital imaging let them see the development and activity of this structure in real time—giving new clues about function and demonstrating that, contrary to previous thinking, multiple new terminal organelles often form before cell division is observed.
From the standpoint of basic science, this research demonstrates the feasibility of using fluorescent proteins to study how organelles in these incredibly tiny bacteria grow and what their functions are. From a medical standpoint, however, they point the way to potential new drug targets and therapies to stop walking pneumonia and chronic bronchitis infections in their tracks.
Since the organelle is involved in colonization of epithelial tissues in human lungs, finding a way to stop such attachment or gliding could halt infections or make them far less severe.
“M. pneumoniae accounts for 20 percent of community-acquired pneumonias in this country,” said Krause. “Finding out more about how the bacterium that causes the disease works gives us a new edge in thinking of ways to overcome such infections.”
Source: University of Georgia