Anti-microbial 'grammar' may mean new medicines

In most languages, sentences only make sense if the words are placed in the right order. Now, MIT researchers and an IBM colleague have used grammatical principles to help their search for new antimicrobial medicines.
After identifying "grammatical" patterns in naturally occurring antimicrobial peptides, the researchers custom-designed molecules that proved extremely effective in killing microbes, including anthrax bacteria. The research could lead to new medicines to combat deadly drug-resistant bacteria.
"In the last 40 years, there have been only two new classes of antibiotic drugs discovered and brought to the market," said graduate student Christopher Loose, lead author of a paper on the work that appears in the Oct. 19 issue of Nature. "There is an incredible need to come up with new medicines."
Loose, research associate Kyle Jensen and Professor Gregory Stephanopoulos of the Department of Chemical Engineering are focusing their attention on antimicrobial peptides, or short strings of amino acids. Such peptides are naturally found in multicellular organisms, where they play a role in defense against infectious bacteria.
The researchers' newly designed peptides were shown to be effective against dangerous microbes such as Bacillus anthracis (anthrax) and Staphyloccus aureus, a bacteria that spreads in hospitals and is frequently drug-resistant. The peptides may also be less likely to induce drug resistance in these bacteria, according to the researchers.
Antimicrobial peptides act by attaching to bacterial membranes and punching holes in them, an attack that is general to many different types of bacteria and is difficult for them to defend against. "There's no quick easy mutation fix for a bacteria to get around this non-specific membrane attack," said Loose.
The peptides are generally short, consisting of about 20 amino acid building blocks. The molecules naturally fold into a helix, with positively charged areas running along one side of the helix and hydrophobic (water-resisting) areas along the other side. The charged ends allow the peptides to latch onto the bacteria by attracting the negative charges of the bacterial membrane, while the hydrophobic ends punch holes in the membrane.
Because there are 20 naturally occurring amino acids, there are about 1026 possible peptide sequences of length 20. Some of those kill microbes with varying levels of effectiveness; the overwhelming majority have no effect.
With such a mind-boggling number of possible combinations, it is extremely difficult to find effective antimicrobial peptides by using traditional methods such as testing random sequences or slightly tweaking naturally existing peptides. "Designing them from scratch is quite difficult," said Loose.
Instead, the researchers decided to take a more strategic approach, based on grammatical patterns in the peptide sequences.
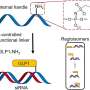
At its essence, a "grammar" is a simple rule that describes the allowed arrangements of words in a given language. As it applies to peptides, the sequence can be thought of as a sentence, while the individual amino acids are the words. For example, the sequence QxEAGxLxKxxK, where x is any amino acid and Q, E, A, etc. are specific amino acids, is a pattern that occurs in more than 90 percent of a certain class of insect antimicrobial proteins known as cecropins.
In this case, the researchers, led by Jensen and Isidore Rigoutsos of IBM Research (Rigoutsos is also a visiting lecturer in the Department of Chemical Engineering), used a pattern discovery tool to find about 700 grammatical patterns in the sequences of 526 naturally occurring antimicrobial peptides.
To design their new peptides, the researchers first came up with all possible 20-amino acid sequences in which each overlapping string of 10 amino acids conformed to one of the grammars. They then removed any peptides that had six or more amino acids in a row in common with naturally occurring peptides. Then, they threw out sequences that were very similar to each other and chose 42 peptides to test.
About half of the peptides displayed significant antimicrobial activity against two common strains of bacteria - Escherichia coli and Bacillus cereus. That is a much higher success rate than one would expect from testing randomly generated sequences, and much higher than the success rate for peptides with the same amino acids as the designed sequences, but in a shuffled order.
"We've been able to focus our shotgun approach so that half of the time, we get a hit," said Loose.
In further tests, two of the designed peptides showed very high effectiveness against two types of especially dangerous bacteria, S. aureus and anthrax.
The researchers have already begun using their technique to further refine the most effective peptides by tinkering with the sequences and altering traits like charge and hydrophobicity. They hope this process will eventually lead to new, more effective antimicrobial medicines.
Source: MIT