When nerve cells can’t make contact
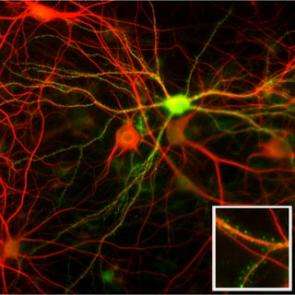
Using an animal model, brain researchers in Göttingen have examined the effects of mutations that cause autism in humans. These are mutations in the genes which carry the building instructions for proteins in the neuroligin family. The study published in the scientific journal Neuron (September 21, 2006) shows that neuroligins ensure that signal transmission between nerve cells functions. In the brain of genetically altered mice without neuroligins, the contact points at which the nerve cells communicate, the synapses, do not mature. The researchers assume that similar malfunctions are experienced by autistic patients.
Autism is one of the most common psychiatric illnesses. Around 0.5 percent of all young children have a syndrome belonging to the "autistic spectrum". The main symptoms of this developmental malfunction are delayed language development or no language development at all, disturbed social behaviour and repetitive behaviour patterns. In many patients, the disease is accompanied by mental disability. Autistic individuals exhibiting high intelligence or outstanding skills in a particular area, called "savants", such as the main character in the film "Rain Man", are rare.
Even up to the middle of the last century, exceptionally cold emotional behaviour on the part of the mother was given as the cause for autism. However, the "refrigerator mom" theory has now been refuted. The belief widely held in the 1990s that the measles, mumps and rubella vaccine could cause autism in small children has no scientific basis at all. Today, it is clear that genetic factors are the major cause of the illness. Studies of identical twins have been particularly convincing in demonstrating this fact - the probability that the identical twin of an autistic person will also be autistic lies between 80 and 95 percent.
In 2003, French geneticist Thomas Bourgeron showed in an investigation of families with several autistic children that mutations in the two genes NLGN3 and NLGN4X had lead to a complete loss of function in the genes and triggered autism in affected patients. Bourgeron’s work sent a shock wave through neuro-scientific institutes worldwide, as the a NLGN genes were not unknown. They are responsible for the creation of two proteins, neuroligin-3 and neuroligin-4, which are considered to play an important part in the structure of nerve cell contacts.
Nerve cells communicate with each other at specialized contact points, the synapses. When stimulated, a transmitting nerve cell emits neurotransmitters. These signal molecules reach the receiving cell and affect its activity status - provided the receiving cell has "aerials" on its synapses - receptors that bind the chemical signals. The scientists speculated that this process could be disrupted if the nerve cells have no neuroligins.
At the time of Bourgeron’s discovery, Nils Brose and Frederique Varoqueaux, brain researchers at the Max Planck Institute for Experimental Medicine in Göttingen, in collaboration with colleagues Weiqi Zhang from the neighbouring University Hospital and US geneticist Thomas Südhof, had already been working on neuroligins for ten years - however in mice, not in humans. "We had even already created mutant mice which, in functional terms, were carrying the same mutations as occur in autistic patients. Our mice were also lacking either neuroligin-3 or neuroligin-4," says Brose. The researchers were in possession of the first genetic animal model for autism.
A study published by Brose, Varoqueaux and Zhang in the specialist journal Neuron has shown that this model exhibited a malfunction in the signal transmission between the nerve cells. With his colleague Varoqueaux, Brose has created a mouse line that not only lacked neuroligin-1 or neuroligin-2, both of which have been associated with autism, but were missing all four known variants of the protein simultaneously. The consequences are accordingly more drastic than with autistic patients, who only have one mutated neuroligin gene. Without any neuroligins, the function of the nervous system breaks down completely and the mutant animals die immediately after birth. However, their nerve cells can be examined in detail. According to Brose, "they deliver important findings not only for brain research in general, but also for the possible causes of autism. Our investigations show that the neuroligins regulate the maturation of the synapses. They ensure that there are sufficient receptor proteins on the synaptic membrane of the receiving cell."
What was initially a pure basic research project has consequently acquired direct relevance to medicine. "What we see in our neuroligin mutants is a more intensified form of the malfunction that occurs in the brain of autistic people," says Brose. "I believe that autism is a disease of the synapses, a synaptopathy." The Max Planck researchers in Göttingen now want to carry out an analysis of the behavioural biology of mutant mice lacking not all of the neuroligins, but just neuroligin-3 or neuroligin-4, as is the case of autistic patients with neuroligin mutations. The relevant mutant mice have been available in the laboratory for a long time, "but we only started analyzing their behaviour with specialists a few months ago," says Brose. The first results look most promising - neuroligin-4 mutant mice obviously have disturbed social and anxiety behaviour. "If we succeed in measuring robust, autism-relevant behavioural changes in our mutant mice, then at least the step to experimental diagnosis and therapy in the animal model will be possible."
From the point of view of the geneticist, the scientists in Göttingen have the best known animal model for autism worldwide. However, there is a limitation: only very few cases of autism are caused by neuroligin mutations and, with few exceptions, nobody knows which genetic defects are present in the abundance of other autism patients.
Citation: Varoqueaux, F., Aramuni, G., Rawson, R.L., Mohrmann, R., Missler, M., Gottmann, K., Zhang, W., Südhof, T.C. and Brose, N., Neuroligins determine synapse maturation and function, Neuron 51, 741-754 (2006)
Source: Max-Planck-Institute for Experimental Medicine