New drug target for immune diseases discovered
Researchers from Mount Sinai School of Medicine have found a new mechanism that explains how certain immune cells are activated to create protective antibodies against infections or pathological antibodies such as those present in autoimmune diseases like lupus and rheumatoid arthritis. The research is published online in the September issue of Nature Immunology.
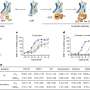
Led by Dr. Andrea Cerutti, MD, Professor of Medicine at Mount Sinai School of Medicine, researchers studied human tissue and immune cells from people with mutations of TACI and MyD88, two proteins required to activate the immune system. MyD88 is a signaling protein that alerts the so-called innate immune system—the immune system encoded at birth that remains unchanged—to the presence of pathogens. TACI is a receptor protein used to activate immune cells in the so-called adaptive immune system, a more sophisticated immune system than the innate, which is dynamic and combats pathogens. These new studies provided important and unexpected new insights in our understanding of immune diseases such as immunodeficiencies and autoimmune disorders.
"Our research shows that TACI and MyD88 are part of an immune circuit that bridges the innate and adaptive immune systems. This circuit makes our immune response more flexible, allowing a more effective generation of protective antibodies during infections. Genetic defects of TACI and MyD88 cause immunodeficiencies characterized by recurrent, life-threatening infections. On the other hand, an abnormally strong TACI-MyD88 interaction may exacerbate autoimmune diseases like lupus or rheumatoid arthritis," said Dr. Cerutti, lead investigator of the study. "Previous studies had suggested an involvement of TACI and MyD88 in lupus. Now that we have identified this interaction, we can figure out a way to inhibit it and prevent these diseases from getting worse."
Autoimmune diseases like lupus and rheumatoid arthritis are characterized by exaggerated production of molecules that activate the adaptive immune system and abnormal antibodies, which attack normal cells causing inflammation and tissue damage. This exaggerated production may occur partly as a result of abnormally strong signaling from TACI via MyD88. By analyzing cells and tissues from immunodeficient patients and genetically engineered mice, Dr. Cerutti's team found a previously unknown interaction between TACI and MyD88 that is important for the production of antibodies against infectious agents. Yet, the same interaction may cause the exaggerated immune response in people with autoimmune diseases.
"Our discovery provides a novel specific target, the signaling pathway between TACI and MyD88, to block the overreaction of the immune system and tissue damage in individuals with autoimmune disorders," said Dr. Cerutti. "We look forward to studying this discovery further and developing therapeutic targets that will inhibit the interaction between TACI and MyD88, preventing autoimmune diseases from progressing with fewer side effects than currently prescribed treatments."
Dr. Cerutti's team collaborated with other researchers at Mount Sinai School of Medicine, including Charlotte Cunningham-Rundles, MD, Professor of Medicine and Pediatrics, and Huabao Xiong, PhD, Assistant Professor of Medicine.
According to the National Women's Health Information Center, autoimmune diseases impact 23.5 million Americans. Common examples include lupus, in which the immune system attacks the skin and/or several organs within the body; rheumatoid arthritis, in which the immune system attacks joints; multiple sclerosis, in which the immune system attacks the nervous system; and Type 1diabetes, in which the immune system attacks insulin-producing cells in the pancreas.