A worm bites off enough to chew (w/ Video)
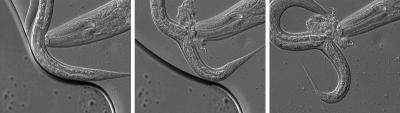
Dramatic scenes are played out under Ralf Sommer's microscope: his research object, the roundworm Pristionchus pacificus, bites another worm, tears open a hole in its side and devours the oozing contents. The squirming victim does not stand a chance in this duel: Caenorhabditis elegans may be a close relative of Pristionchus; unfortunately, however, it does not have the same strong "teeth".
Pristionchus' impressive hunting technique though is not the focus of interest for the biologists from the Max Planck Institute for Developmental Biology in Tubingen. Their concern is the development of its mouthparts. Gilberto Bento and Akira Ogawa from Sommer's team have discovered the control mechanism that lies behind the development of the organism's mouth: if the worm grows up with an abundant supply of bacteria as its source of nutrition, it only develops very small teeth and a narrow oral cavity. If it experiences a lack of food or a high population density at the larval stage, it develops a wide mouth equipped with strong teeth-like denticles. (Nature July 1, 2010)
Worms with wide mouths do not differ genetically from their narrow-mouthed counterparts: "Environmental factors dictate the kind of mouthparts formed by roundworms," says Ralf Sommer, Director of the Department of Evolutionary Biology at the Max Planck Institute for Developmental Biology in Tübingen. If there is a shortage of food during a sensitive phase in larval development, almost all of the worms develop a wide and heavily-armed mouth.
One of the worms' pheromones also triggers the same effect. When this pheromone is present in high concentrations, it signals the presence of high population density. In both cases - that is, food shortage and overpopulation - a signalling pathway specific to the worm is activated, which results in the development of strong teeth, thereby enabling predatory behaviour. This signalling pathway is already known to the researchers: the hormone dafachronic acid and its receptor also ensure that, in times of scarcity, the worms do not develop into adult individuals but remain in a dormant larval state until the environmental conditions improve again.
"Pristionchus' mouth dimorphism demonstrates two fascinating evolutionary principles simultaneously," says Sommer. First, it shows how frugally evolution works: signalling pathways that have already been established are re-used in a new context - biologists refer to this process as co-option. In order to assign a new significance to a signalling chain, all that needs to be done is to activate it at a different time or with a different concentration of the signalling molecule that triggers its activation, as occurs in this case. Moreover, the existence of alternative body structures is viewed as paving the way for evolution: "In order to change the mouth structure permanently, the genetic control would only have to be decoupled from the environmental dependency," explains Ralf Sommer.
Whether Prisionchus' stronger mouth structure is better suited to the hunting of other worms or consumption of fungi, is something the Tübingen scientists have only been able to speculate about thus far. "The fact that the mouth dimorphism became established in the course of evolution would suggest that it offers an important advantage in the natural world," says Ralf Sommer.
More information: Gilberto Bento, Akira Ogawa, Ralf Sommer, Co-option of the hormone-signaling module Dafachronic Acid-DAF-12 in nematode evolution. Nature, July 1, 2010
Provided by Max-Planck-Gesellschaft















