Sluggish cell division may help explain genital defects
Scientists have learned how a gene widely known for precisely positioning and sculpting various organs also controls the speed of cell division, a finding that could be useful for understanding the explosive growth of cancer cells or why increasing numbers of children are being born with genital and urinary tract malformations.
Writing today (Tuesday, June 1) in Nature Communications, researchers at the University of Florida say a gene memorably named Sonic hedgehog controls genital development by regulating a process known as the cell cycle -- a biological event that regulates when, and how fast, cells divide to form hearts, brains, limbs and all the other complex structures needed to build an individual.
The findings in mice provide insight into the molecular mechanisms that underlie growth of urinary and reproductive organs in both sexes. Abnormalities of the genitalia and urinary tract are among the most common birth defects, according to the March of Dimes. Similarly, the ability of Sonic hedgehog to alter the time it takes to complete the cell cycle might also influence tumor growth in a wide range of cancers, including the most common form of skin cancer.
"The role of Sonic hedgehog during embryonic development is to set up the positional addresses of cells in everything from limbs to the spinal cord, telling cells where they are located and what they will become -- a process known as patterning," said senior author Martin Cohn, a Howard Hughes Medical Institute early career scientist and a member of the UF Genetics Institute and the College of Medicine. "We've shown Sonic hedgehog also controls organ growth by determining how long a cell spends preparing to replicate its DNA. The surprise is to find out how much patterning and growth are intertwined. An embryo has only a fixed amount of time to grow. Once we discovered that inactivation of Sonic hedgehog slowed down the cell cycle, it explained the big differences in growth and the structural defects we were finding in genitalia."
The knowledge may help scientists understand why an increasing number of boys are being born with birth defects called hypospadias, which involve incomplete formation of the urethral tube, resulting in an abnormally placed urethral opening on the underside of the penis. About one in 250 children has a urethral tube defect, more than double the frequency of 30 years ago.
The cell cycle controls whether a cell continues to give rise to more cells or stop dividing and become specialized with a specific function to carry out. Humans begin life as a single fertilized egg cell that eventually gives rise to countless cells in an adult. As each cell divides it must proceed through a growth phase, replicate its DNA and divide again, or it can be instructed to stop dividing and perform a specific function.
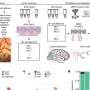
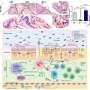
When scientists deleted the Sonic hedgehog gene in specific tissues at different stages of external genital development, they discovered the cell cycle takes longer than it normally does -- about 14.4 hours instead of the usual 8.5 hours for these cells. As a result, fewer cells are produced and genital growth is reduced by about 75 percent. The shape of the genitals is also altered.
"In this case, embryos wind up with underdeveloped, malformed genitalia, and the reason is that it takes nearly twice as long to complete the cell cycle, limiting the number of cells available to build the structure. What is surprising is that the number of cells seems to underlie the shape," said Ashley Seifert, a postdoctoral associate in the department of biology in the College of Liberal Arts and Sciences and first author of the paper. "We did not just see a miniaturized version of the genitals, we observed patterning defects, from subtle changes to severe malformations."
Researchers had thought malformations might be explained because important shaping genes that sculpt the genitalia would be controlled by Sonic and thus turned off in its absence, Seifert said. But instead, scientists found many of these key genes were still expressed in the right places, forcing the scientists to look elsewhere for the cause of the defects. The search led them to the cell cycle.
Employing a technique known as stereology, which is a way to accurately estimate the number of cells in a region by taking smaller samples -- similar to opinion polling -- scientists found decreased cell numbers in the Sonic hedgehog-depleted genitalia.
"When we began, the thought was that these cells may not be dividing at all, but the possibility existed that the cell cycle was just taking too long. This would mean that new cells could just be missing the next exquisitely timed signal required for further growth or patterning," said co-author Brandi Ormerod, a neuroscientist and biomedical engineer in the J. Crayton Pruitt Family department of biomedical engineering with the UF College of Engineering. "Essentially we used methods that we employ frequently in neuroscience to label dividing cells in the brain, determine how many of them there are within a structure, and figure out how long the cell cycle is taking -- we just applied them to a different system during development."
Ormerod, who is also a member of the UF Genetics Institute and the UF Shands Cancer Center, said the cells in developing genitalia may be missing time-sensitive signals that trigger completion of their division when Sonic hedgehog signaling is disrupted.
"The findings point out that Sonic hedgehog may be a very robust agent that induces cell division," said Dr. Susan Mackem, head of the Regulation of Vertebrate Morphogenesis Section at the National Cancer Institute, who was not involved in the study. "Clearly there may be a role for aberrant Sonic hedgehog activity in some forms of cancer. It has been turning up in a number of contexts, which makes sense if it is regulating several components of the cell cycle. You can imagine that if you have a growing population of cells that contains mutations, hyperactivity in the Sonic hedgehog signaling pathway will have a profound effect on tumor growth."