Physics of gene transcription unveiled
(PhysOrg.com) -- A research team has made precise measurements of where and how RNA polymerase encounters obstacles while it reads nucleosomal DNA.
How single DNA molecules are "unzipped" by enzymes to make copies of genetic information is not just a biochemistry problem -- it's a physics problem, too.
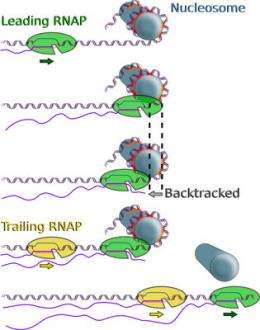
That's because DNA molecules, often illustrated as long, wispy double helixes, are usually wound and packed into units called nucleosomes. For RNA polymerase (RNAP), an enzyme that "reads" the DNA, to get to the genetic material, it needs to unwind this tightly wound spool -- and along the way, it encounters obstacles on which it needs to exert some force.
These impossibly subtle forces are what Michelle Wang, professor of physics and Howard Hughes Medical Institute investigator, and colleagues have uncovered with precision down to nanometers. Publishing online in Nature Structural and Molecular Biology May 9, her team has made precise measurements of where and how RNAP encounters obstacles while it reads nucleosomal DNA. They have shown that when RNAP molecules work as partners, they have better access to the DNA.
"When RNAP encounters a nucleosome, it is not fully understood what happens," Wang said. "How strong is the barrier? Where does the polymerase pause?"
The researchers have answered these questions using various biochemical methods and a DNA unzipping technique invented in Wang's lab that uses an optical trap. This harnesses a single strand of DNA attached to a refractive sphere for manipulation.
The scientists showed exactly where the RNAP pauses along the DNA -- accurate to within a few base pairs, which are the nucleotides along opposite sides of the DNA. They also discovered that the pauses are caused in part by RNAP backtracking slightly when encountering an obstacle. Finally, they have provided evidence that two RNAP molecules working in tandem can help overcome obstacles.
"Think of the DNA sequence as a road," Wang said. "The DNA sequence gives you a sense of where the road marks are. As the polymerase moves, I want to pin down the exact location of where it is. Unzipping DNA is a powerful tool that actually allows us to measure the location of an RNAP to within a couple of base pairs."
The paper's first author is Jing Jin, a postdoctoral associate in Wang's lab, and includes contributions from former graduate students Lu Bai and Daniel S. Johnson; Cornell senior lecturer Robert M. Fulbright; and collaborators from the National Cancer Institute's Center for Cancer Research.
Provided by Cornell University