Synthetic lethality: A new way to kill cancer cells
Ovarian and breast cancer treatments being developed that mix a protein inhibitor and traditional anticancer drugs are showing signs of success, according to a new review for Faculty of 1000 Biology Reports.
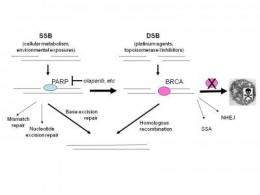
Susan Bates and Christina Annunziata looked at several recent papers on this form of treatment, which takes advantage of the synthetic lethality of BRCA (breast cancer susceptibility genes) and poly-ADP ribose polymerase (PARP) proteins to attack cancerous cells whilst sparing healthy ones.
BRCA and PARP are two key players in DNA repair and have different but complementary functions in the cell. Loss of the BRCA protein still allows the cell to survive but greatly increases its chances of becoming cancerous through the accumulation of mutations. The loss of both proteins, however, kills the cell in a process called synthetic lethality.
Researchers, by using drugs to block the activity of PARP in cells missing BRCA, such as those found in certain breast and ovarian cancers, can help spare healthy, non-cancerous cells because they have functional BRCA and are not affected by the loss of PARP. Thus, only cancer cells without functional BRCA protein are killed by drugs that inhibit PARP.
Recent clinical trials have shown that cancers caused by mutations that knock out BRCA activity can be controlled by blocking PARP activity with specific drugs. Patients were treated with traditional anticancer drugs alone or in combination with one of two new PARP inhibitors, olaparib or BSI-201.
Bates notes that patients on combination therapy had improved "[disease] progression-free survival, and overall survival" as compared to patients treated with traditional drugs alone.
Bates is optimistic about the promise of combining PARP inhibitors with existing cancer drugs. She says that the results of these clinical trials "have provided proof of principle in achieving synthetic lethality" with PARP-inhibiting drugs and that treatments combining novel PARP inhibitors with traditional chemotherapeutic drugs have the potential to vanquish BRCA-associated breast and ovarian cancers.
More information: The full text of this article is available for subscribers at f1000biology.com/reports/10.3410/B2-10/ or for reporters at faculty1000.files.wordpress.co … /02/bates-report.pdf















