Within a cell, actin keeps things moving
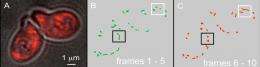
(PhysOrg.com) -- Using new technology developed in his University of Oregon lab, chemist Andrew H. Marcus and his doctoral student Eric N. Senning have captured what they describe as well-orchestrated, actin-driven, mitochondrial movement within a single cell.
That movement -- documented in a paper appearing online the week of Dec. 14-18 ahead of regular publication in the Proceedings of the National Academy of Sciences -- appears to be coordinated by mitochondria's recruitment of actin-related proteins that rapidly assemble into extended fractal-like structures in a molecular chemical reaction known as polymerization. The coordinated movement of mitochondria is important for reproduction of identical daughter cells, and the sorting of mitochondrial DNA into the spinoff cells.
The research was done with a molecular fluorescence technology called Fourier imaging correlation spectroscopy that allows researchers using focused laser beams to see, measure and map the intermittent movement of mitochondria at micron scales. Marcus will discuss the technology, developed with funding from the National Institutes of Health and National Science Foundation, at the 2010 annual meeting of the American Physical Society in Portland, Ore., in March. It also was detailed in a paper published online in October by the journal Annual Reviews of Physical Chemistry.

In their project published by PNAS -- funded by the NIH -- Senning and Marcus looked at actin's behavior using inhibitory agents to monitor mitochondrial activity in Saccharomyces cerevisiae, a species of budding yeast often used in research. They also introduced two defective forms of the protein. Their technique included the use of hormones to trick a yeast cell into thinking it was about to mate, so that it stops dividing and sits and fluctuates -- much like a car in idle. From this state, the images are drawn.
The picture that emerged, Marcus said, was that actin is drawn to the surfaces of mitochondria to regulate the polymerization machinery so that it operates in an efficient, organized manner. The findings, the researchers wrote, lend support to an existing model in which non-equilibrium forces are directly coupled to mitochondrial membrane surfaces. In effect, the findings support the idea that despite the cramped quarters of molecules in densely packed cells, intracellular transport is accomplished by coordinating the movements of a multi-faceted machine, rather than resulting from random (Brownian) movements of material based on what obstacles will allow.
The quest for understanding the machinery is more than just biological, where this research provides insight into how the cell moves its mitochondria into the daughter cells, Marcus said. The knowledge could become useful in the production of nanotechnology devices.
"A central question in modeling cell transport is whether the cytoplasm may be viewed as a simple extension of a complex fluid at equilibrium or if non-equilibrium effects dominate the motions of intracellular species," he said. "If somebody wants to design a micron-scale machine or make a self-replicating device, one would have to have these physical principles in place. One would need to have a motor in place and know how much force the motor needs to apply, either cooperatively or individually with other components."
Provided by University of Oregon