Thermochemical nanolithography now allows multiple chemicals on a chip
(PhysOrg.com) -- Scientists at Georgia Tech have developed a nanolithographic technique that can produce high-resolution patterns of at least three different chemicals on a single chip at writing speeds of up to one millimeter per second. The chemical nanopatterns can be tailor-designed with any desired shape and have been shown to be sufficiently stable so that they can be stored for weeks and then used elsewhere. The technique, known as Thermochemical Nanolithography is detailed in the December 2009 edition of the journal Advanced Functional Materials. The research has applications in a number of scientific fields from electronics to medicine.
"The strength of this method is really the possibility to produce low-cost, high-resolution and high-density chemical patterns on a sample that can be delivered in any lab around the world, where even non experts in nanotechnology can dip the sample in the desired solution and, for example, make nano-arrays of proteins, DNA or nanoparticles," said Elisa Riedo, associate professor in the School of Physics at the Georgia Institute of Technology.
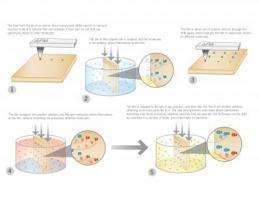
Conceptually, the technique is surprisingly simple. Using an atomic force microscope (AFM), researchers heat a silicon tip and run it over a thin polymer film. The heat from the tip induces a local chemical reaction at the surface of the film. This reaction changes the film's chemical reactivity and transforms it from an inert surface to a reactive one that can selectively attach other molecules. The team first developed the technique in 2007. Now they've added some important new twists that should make thermochemical nanolithography (TCNL) an extremely useful tool for scientists working at the nanoscale.
"We've created a way to make independent patterns of multiple chemicals on a chip that can be drawn in whatever shape you want," said Jennifer Curtis, assistant professor in the School of Physics.
Being able to create high-resolution features of different chemicals in arbitrary shapes is important because some nanolithography techniques are limited to just one chemistry, lower resolutions and/or fixed shapes. In addition, TCNL's speed capability of one millimeter per second makes it orders of magnitude faster than the widely used dip-pen nanolithography, which routinely clocks at a speed of 0.0001 millimeters per second per pen.
The research is enabled by heated AFM probe tips that can create a hot spot as small as a few nanometers in diameter. Such tips are designed and fabricated by collaborator Professor William King at the University of Illinois, Urbana-Champaign. "The heated tip allows one to direct nano-scale chemical reactions," said King.
The new technique produces multiple chemical patterns on the same chip by using the AFM to heat a polymer film and change its reactivity. The chip is then dipped into a solution, which allows chemicals (for example, proteins or other chemical linkers) in the solution to bind to the chip on the parts where it has been heated. The AFM then heats the film in another spot. The chip is dipped into another solution and again another chemical can bind to the chip.
In the paper, the scientists show they can pattern amine, thiol, aldehyde and biotin using this technique. But in principle TCNL could be used for almost any chemical. Their work also shows that the chemical patterns can be used to organize functional materials at the surface, such as proteins and DNA.
"The power of this technique is that in principle it can work with almost any chemical or chemically reactive nano-object. It allows scientists to very rapidly draw many things that can then be converted to any number of different things, which themselves can bind selectively to yet any number of other things. So, it doesn't matter if you're interested in biology, electronics, medicine or chemistry, TCNL can create the reactive pattern to bind what you choose," said Seth Marder, professor in Tech's School of Chemistry and Biochemistry and director of the Center for Organic Photonics and Electronics.
In addition, TCNL allows the chemical writing to be done in one location with the nano-object patterning in another, so that scientists who aren't experts in writing chemical patterns on the nanoscale can still attach their objects to it. It's the technique's stability that makes this possible.
"Once you draw the pattern, it's very stable and non-reactive. We've shown that you can have it for more than a month, take it out and dip it and it still will bind," said Riedo.
"I would like to think that several years from now people will have access to a TCNL tool that enables them to do this patterning at a place like Georgia Tech, that's much less expensive than the kind of nanolithography tools we currently use in our clean room," said Marder.
Provided by Georgia Institute of Technology

















