NIST finds 'a touch of glass' in metal, settles century-old question
Better predictions of how many valuable materials behave under stress could be on the way from the National Institute of Standards and Technology (NIST), where scientists have recently found evidence of an important similarity between the behavior of polycrystalline materials—such as metals and ceramics—and glasses.
Most metals and ceramics used in manufacturing are polycrystals. The steel in a bridge girder is formed from innumerable tiny metal crystals that grew together in a patchwork as the molten steel cooled and solidified. Each crystal, or “grain,” is highly ordered on the inside, but in the thin boundaries it shares with the grains around it, the molecules are quite disorderly. Because grain boundaries profoundly affect the mechanical and electrical properties of polycrystalline materials, engineers would like a better understanding of grain boundaries’ formation and behavior. Unfortunately, grain boundary formation in most technically useful alloys has eluded efforts to observe it for a century.
“You’d like to have simple engineering rules regarding how a material’s going to break,” says NIST materials scientist Jack Douglas. “For example, corrosion typically travels along grain boundaries, so polycrystals usually fracture along them. But metals melt and deform at very high temperatures, so observing them under those conditions is a challenge.”
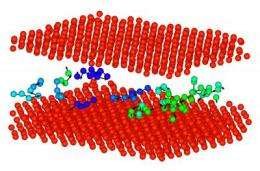
While some scientists had speculated that the molecules in grain boundaries behave similarly to the way molecules do in glass-forming liquids, whose properties are well understood, none had found conclusive evidence to back up such a claim. That started to change when NIST theorist James Warren saw a conference presentation by the University of Alberta’s Hao Zhang concerning some odd “strings” of atoms in his simulation of grain boundary motion using a simulation technique called molecular dynamics. The collective atomic behavior observed in grain boundaries reminded the team of prior findings made at NIST about glass-forming liquids, whose atoms also form strings.
Subsequently, the team showed that the strings of atoms arising in grain boundaries are strikingly similar in form, distribution and temperature dependence to the string-like collective atomic motions generally found in glass-forming liquids—and that properties for both types of substances change with temperature in virtually the same way. “This work represents a paradigm shift in our understanding of grain boundaries,” Douglas says. “All the important qualities relating to atomic motion in both of these types of materials—the development of these string-like atomic motions, or the amplitude at which their atoms rattle—are strikingly similar. For all intents and purposes, grain boundaries are a type of glass.”
Douglas says the findings could permit substantial progress in predicting the failure of many materials important in construction and manufacturing and could improve our understanding of how crystals form boundaries with one another.
Citation: H. Zhang, D.J. Srolovitz, J.F. Douglas and J.A. Warren. Grain boundaries exhibit the dynamics of glass-forming liquids. Proceedings of the National Academy of Sciences. Vol.106, No. 10 (2009).
Source: National Institute of Standards and Technology (NIST)

















