First high-resolution images of bone, tooth and shell formation

Researchers at Eindhoven University of Technology (The Netherlands) have for the first time made high-resolution images of the earliest stages of bone formation. They used the world's most advanced electron microscope to make three-dimensional images of the nano-particles that are at the heart of the process. The results provide improved understanding of bone, tooth and shell formation. For industrial applications, they promise better materials and processes based on nature itself. The findings form the cover story of Science magazine's Friday 13 March edition.
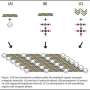
Led by dr. Nico Sommerdijk, the researchers imaged small clusters with a cross-section of 0.7 nanometer in a solution of calcium carbonate (the basic material of which shells are made). They showed for the first time that these clusters, each consisting of only about ten ions, are the beginning of the growth process through which the crystalline biomineral is ultimately formed.
To do this they used the very high resolution of a special electron microscope: the cryoTitan (of FEI Company). This enabled them, as the first in their field of research, to make three-dimensional images of very rapidly frozen samples. These showed how the clusters in the solution nucleate into larger, unstructured nano-particles with an average diameter of around thirty nanometers.
An organic surface applied by the researchers ensures that these nano-particles can grow into larger particles, in which crystalline regions can later form by ordering of the ions. The TU/e researchers also demonstrated a second function of the organic layer: it controls with great precision the direction in which the mineral can grow into a fully fledged biomineral. They now hope to show that the mechanism they have identified also applies to the formation of other crystalline biominerals, and perhaps even to other, inorganic materials.
This is important for research into bone growth and bone-replacement materials. In addition it could be used in nanotechnology, to allow the growth of nano-particles to be controlled in the same way as seems to be the case in nature: through subtle interactions between organic and inorganic materials.
About biomineralization
Biomineralization is the formation of inorganic materials in a biological environment, as it is found in bones, teeth and shells. In this process the formation of the mineral is controlled with great precision by specialized organic biomolecules such as sugars and proteins. Although the underlying mechanisms have already been studied for a long time, the process is still not fully understood.
A widely used strategy is the use of so-called biomimetic studies, in which the process of biomineralization is simulated by a simplified system in a laboratory. This allows parts of the mineralization process to be studied individually.
With this approach and by using the unique electron microscope referred to above, Sommerdijk's research group in the Chemical Engineering and Chemistry department at TU/e have been able to image the earliest stages of such a biomimetically controlled mineralization reaction.
More information: Science, 13 March 2009, "The initial stages of template-controlled CaCO3 formation revealed by Cryo-TEM"; Emilie M. Pouget, Paul H.H. Bomans, Jeroen A.C.M. Goos, Peter M. Frederik, Gijsbertus de With and Nico A.J.M. Sommerdijk.
Source: Eindhoven University of Technology