Genetic change extends mouse life, points to possible treatment for ALS
There are many ways to die, but amyotrophic lateral sclerosis (ALS), also known as Lou Gehrig's disease must be one of the worst. By the time a patient notices muscle weakness, the neurons that control the muscles have already begun dying, in an untreatable process that brings death within two to five years.
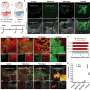
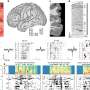
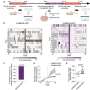
In a series of experiments reported today in the Journal of Neuroscience, a University of Wisconsin-Madison pharmacy researcher was able to prolong life and slow nerve deterioration in a mouse with a genetic form of ALS, or amyotrophic lateral sclerosis. Marcelo Vargas, a postdoctoral fellow in the laboratory of Jeff Johnson, professor in the School of Pharmacy, tested mice that carried an extra gene that pushed support cells for the neurons into overdrive, causing them to pump out extra quantities of the anti- oxidant glutathione.
The gene in question, called Nrf2, has long been a research focus for Johnson, who is also a Waisman Center investigator.
Although oxidation is a major cause of cell death in Parkinson's disease and Alzheimer's disease as well as ALS, antioxidant treatments have failed to slow these diseases.
But the mice with extra copies of Nrf2 produced glutathione right alongside the vulnerable neurons, and that made all the difference, says Johnson. These special mice were engineered in collaboration with Albee Messing, a professor in the UW-Madison School of Veterinary Medicine and also an investigator at the Waisman Center. "It's extremely difficult to increase glutathione in the central nervous system," Johnson says. "You can't just shoot it into people or animals. But we found a 25 percent increase in the molecule in the spinal cords."
Although the mice did eventually die of ALS, they lived longer, and the disease appeared 17 days later than in mice that lacked the extra Nrf2 gene, Johnson says. "This was a very aggressive model of ALS, so a life extension of 21 days is thought to be pretty significant, roughly equivalent to five to 10 years in human patients."
The inserted Nrf2 gene was only active in support cells called astrocytes, which promote health among the neurons that actually carry nerve signals, Johnson explains. "We have taken this normal function of producing antioxidants and added to it. It's like putting the astrocytes on steroids."
Experiments performed on mouse astrocytes and nerve cells in a dish confirmed the source of the protection, Johnson adds. "We can completely reverse the toxicity of the sick astrocytes. The mutated protein that causes ALS is still there, but Nrf2 makes glutathione that completely blocks it."
Although the mice that Johnson tested carried the inherited form of ALS, most patients do not have an inherited disease. However, Johnson says the mice are still a good testbed for ALS treatments. "The endpoints that we are blocking, including death of neurons and separation of neurons from the muscle, are seen in all forms of ALS; that's what makes this so exciting. We are not targeting the mutant protein that causes the disease; we are targeting the astrocytes' mechanism that preserves the neurons. The mutant protein is still in all cells of the spinal cord; we are just over-expressing the Nrf2 gene — causing it to make more glutathione — and that provides the protection."
Nrf2 activates a system, or pathway, that is also attracting interest from researchers working on Parkinson's disease, Alzheimer's disease, Huntington's disease and stroke, says Johnson. "It's been exciting. This hypothesis came to me when I was in graduate school in 1990, and this year it seems to be coming to fruition" as a possible treatment for several neurodegenerative diseases.
The results may be promising, but inserting genes, or gene therapy, has had few successes to date. Yet by positively identifying the chemical pathway that keeps neurons healthy in ALS, Johnson is in a position to search for a drug that can enter the brain and activate the Nrf2 system. That quest is already under way at an automated screening facility at UW-Madison, where more than 50,000 molecules are being tested for their ability to activate Nrf2.
Years of research will be needed before today's results can be translated into a treatment for patients, however. "If everything worked perfectly, we probably could have something in two to three years, but the odds of that happening are pretty low," says Johnson. "But in five to eight years, I hope to have something can get through all the regulatory hoops."
Finding that one mechanism is involved in several neurodegenerative diseases is significant, Johnson says, because it attracts a broader group of scientists and funders to the work. "Something seems to be going wrong with the neurons because the astrocyte does not function right. Now, it looks like we have the potential to fix the astrocyte, so it can preserve the neurons for a longer period."
Source: University of Wisconsin-Madison