When less is more: Brief inhibition of cancer target is effective and less toxic
New research shows that the delicate balance between maximum clinical impact and toxicity may not be quite as fragile as scientists had previously believed. The study, published by Cell Press in the December issue of the journal Cancer Cell, is likely to have a major impact on the future design and implementation of targeted cancer therapies.
Small molecule tyrosine kinase inhibitors, such as imatinib (Gleevec) which targets the BCR-ABL kinase associated with chronic myeloid leukemia (CML), have prolonged half-lives in patients and provide almost continuous inhibition of their targets when administered once daily. It was generally assumed that prolonged target inhibition was required to achieve clinical activity with such inhibitors. This assumption was a major driving force in both drug discovery efforts where many agents with short half-lives were abandoned prior to clinical development and also in the design of dosing schedules.
Desatinib is a second generation BCR-ABL kinase inhibitor with greater potency but a much shorter half-life than imatinib and all other approved kinase inhibitors. "The clinical development of dasatinib initially proceeded using a twice-daily dosing schedule with the goal of providing continuous target coverage," explains lead study author, Dr. Neil P. Shah from the University of California, San Francisco School of Medicine. Dr. Shah was the lead author of a clinical study, recently published in the Journal of Clinical Oncology, which found that the clinical benefit of dasatinib was equivalent when dosed once daily, and importantly, tolerability was superior to the twice-daily schedule.
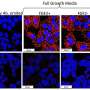
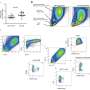
Dr. Shah and colleagues sought to determine the relationship between the depth and duration of kinase inhibition with respect to killing cancer cells, and to begin to characterize the molecular mechanism linking potent transient kinase inhibition with cancer cell death. The researchers found that, as expected, potent inhibition of BCR-ABL kinase activity is transiently achieved in CML patients treated with dasatinib once daily and that maximal clinical benefit is associated with maximal BCR-ABL inhibition. Transient inhibition of BCR-ABL was also effective against CML cells grown in the lab.
Significantly, transient inhibition of a separate kinase was effective in a lung cancer cell line which suggests that potent transient kinase inhibition may be effective in a number of other cancers, and that a cancer cell's "addiction" to a continuous specific growth-promoting signal can be exploited therapeutically.
The researchers went on to demonstrate that the kinetics of cancer cell death and activation of the cell-death associated protein BIM were identical for transient and prolonged target inhibition therapies. These findings challenge the assumption that maximum clinical impact requires continuous inhibition of cancer targets. "Our results provide compelling rationale for the clinical development of compounds capable of achieving potent kinase inhibition, irrespective of biological half-life," concludes Dr. Shah. "It should be possible to optimize individual patient doses, balancing target inhibition with toxicity, in pursuit of the goal of rational personalized cancer medicine."
Source: Cell Press