Research reveals mechanism linking serotonin with regulation of food intake
Genetic mouse models have provided surprising insight into mechanisms linking serotoninergic compounds with the regulation of feeding behavior and body weight. The research, published by Cell Press in the November 26th issue of the journal Neuron, pinpoints a specific group of brain cells that mediate energy balance and may lead to the development of antiobesity drugs with fewer side effects.
The 5-hydroxytryptamine (5-HT) system, which includes the 5-HT2C receptor (5-HT2CR) subtype, has been shown to play an important role in the regulation of energy homeostasis. Previous work demonstrated that excessive food intake and obesity are linked with 5-HT2CR deficiency and that the atypical antipsychotic drugs, which appear to block 5-HT2CRs, are associated with serious weight gain. Further, 5-HT2CRs are known to contribute to the appetite suppressant effects of d-fenfluramine, a drug widely prescribed to combat obesity in the 1990s that was later banned because of a negative impact on the heart.
"We have known for some time that drugs activating 5-HT2CRs potently suppress appetite, but the underlying mechanisms for these effects are not fully understood," says senior study author Dr. Joel K. Elmquist from the University of Texas Southwestern Medical Center at Dallas. "We also know that pro-opiomelanocortin (POMC) neurons in the hypothalamus of the brain release neuropeptides that activate the central melanocortin receptors which is required to maintain food intake, body weight and glucose homeostasis. The melanocortin pathway has been hypothesized to be a downstream mediator of the effects of 5-HT2CRs on feeding behavior."
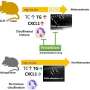
To investigate whether 5-HT action on POMC neurons is sufficient to mediate the inhibitory effects of 5-HT compounds on appetite, Dr. Elmquist and colleagues generated mice in which 5-HT2CR was globally disrupted (called 2C null), but 5-HT2CR expression could be reactivated in any cells that were engineered to express Cre-recombinase. Crossing 2C null mice with mice that express Cre-recombinase in POMC neurons resulted in mice with 5-HT2CRs expressed only in POMC neurons (called 2C/POMC). The researchers observed that 2C null mice, as expected, exhibited excessive eating, hyperactivity, and obesity and showed reduced responses to 5-HT drugs known to suppress appetite. However, unexpectedly, all the metabolic deficiencies observed in the 2C null mice were completely restored in the 2C/POMC mice.
These findings suggest that expression of 5-HT2CRs solely on POMC neurons is sufficient to mediate the effects of serotoninergic compounds on food intake. "Our unique genetic mouse models have revealed that POMC neurons are physiologically important targets of potent anorexigenic 5-HT compounds such as d-fenfluramine to suppress appetite. In addition, our results highlight the importance of the central 5-HT2CRs expressed by POMC neurons in maintaining normal feeding behavior and body weight homeostasis," concludes Dr. Elmquist.
Interestingly, another characteristic phenotype of 2C null mice, epileptic seizures, was not rescued by re-expression of 5-HT2CRs on POMC neurons. These findings indicate that 5-HT2CRs regulate neuronal excitability through actions in other CNS sites, and the re-activatable 2C null mouse model would serve as an excellent tool to tackle this issue as well as to reveal other physiological functions of 5-HT2CRs.
Source: Cell Press