Maternal Obesity Can Program Fetal Brain to Induce Adult-onset Obesity
(PhysOrg.com) -- Researchers at the University at Buffalo have found that fetuses of obese mother rats were programmed in utero to develop obesity in adulthood.
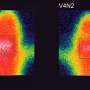
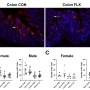
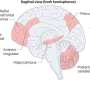
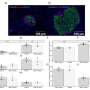
Moreover, they have shown for the first time that the metabolic programming occurs in the fetal hypothalamus, the area of the brain responsible for maintaining the body's energy homeostasis (body weight) throughout life.
Levels of the hormones insulin and leptin also were elevated in fetuses of these obese mother rats, abnormalities that have been correlated with increased appetite and insulin resistance (a prelude to diabetes), as well as obesity and hypertension.
"Our earlier studies looked at newborn rats of the obese mothers in the post-weaning period, so we didn't know how early this programming occurred," said Mulchand Patel, Ph.D., UB Distinguished Professor of Biochemistry and senior author on the study. "Now we know it occurs in utero and specifically in the hypothalamus.
"While these studies were done with rats, there is good reason to think the mechanism would be similar in humans," he said. " The fact that more than one-third of women of child-bearing age in the United States are expected to be overweight or obese during pregnancy, based on a 2003 study, does not portend well for good health of their offspring."
The new findings were published in the October 2008 issue of the American Journal of Physiology, Endocrinology and Metabolism. Malathi Srinivasan, Ph.D., research scientist in the UB Department of Biochemistry, is first author.
Metabolic programming, sometimes called dietary patterning, isn't a new phenomenon, Patel noted. "Epidemiologic studies of malnourished mothers showed that their babies often were underweight and at increased risk for several chronic diseases as adults. Animal studies on maternal protein malnourishment or caloric restriction have shown that pre- and immediate postnatal nutritional modifications have long-term consequences on adult-onset diseases."
Patel and colleagues are pioneers in the investigation of metabolic programming effects when the composition of the diet is manipulated -- changing the percentages of carbohydrates and fat, while keeping calories constant.
Patel reported evidence of metabolic programming in 2002, when his laboratory showed that in rats, consumption of a milk formula high in carbohydrates during the critical early weeks of postnatal life caused permanent changes in pancreatic islets, leading to overproduction of insulin and development of obesity in adulthood.
"Metabolic signals are reset in response to a high carbohydrate milk formula given to newborn rats during the suckling period, which induces permanent changes at the molecular level in our rat model," Patel said. "The HC (high carbohydrate) phenotype is maintained for life and is spontaneously transmitted to succeeding generations."
Unlike many models investigating the role of maternal obesity on their offspring, the mother rats used in this study consumed normal laboratory chow during pregnancy, Paten noted.
"Our findings that malprogramming effects induced during fetal development in the altered intrauterine environment in obese mother rats predispose the offspring for adult-onset obesity underscore the importance of women maintaining optimal conditions during their pregnancies," said Patel.
Additional contributors to the study were Catherine Dobbs and Tao Gao, from the UB Department of Biochemistry; Hasam Ghanim and Paresh Dandona, from the UB Department of Medicine; and Peter J. Ross and Richard W. Browne, from the UB Department of Biotechnical and Clinical Laboratory Sciences -- all part of UB's School of Medicine and Biomedical Sciences.
Ghanim and Dandona also are affiliated with Kaleida Health's Diabetes and Endocrinology Center of Western New York.
Patel's research is supported in part by grants from the National Institutes of Health.
Provided by University at Buffalo