Duke innovations improve accuracy of MRI as internal 'thermometer'
Duke University chemists say they have developed a new way to measure temperature changes inside the body with unprecedented precision by correcting a subtle error in the original theory underlying Magnetic Resonance Imaging (MRI).
"We can get five to 10 times better accuracy in temperature maps than is possible with the best possible conventional methods," said Warren Warren, a Duke chemistry professor who is corresponding author of a new report appearing in the Oct. 17 issue of Science. The work was funded by the National Institutes of Health.
The new technique "is suitable for imaging temperature in a wide range of environments," added the report.
MRI is a radiation-free technology for imaging patients' interior anatomies. It works because hydrogen atoms in internal organs will broadcast their locations when subjected to selected radio waves in the presence of a strong, computer-programmable magnetic field.
MRI scans can also be used to estimate interior temperature changes in procedures like hyperthermia cancer therapy, where focused heat is used to kill internal tumors. This is because the hydrogen atoms in water shift their MRI broadcasting frequencies in a predictable way as water temperatures change. And water is a major component of molecules in bodily tissues.
Though precise in evaluating water temperature changes in isolation, conventional MRI works imperfectly as an internal thermometer within actual patients. That's because the magnetic field's interactions with hydrogen atoms vary widely within patients' bodies, and those interactions also shift from minute to minute, Warren said.
"Current methods break down in the very systems that are of greatest interest, those that are inhomogeneous and that change with time," the report said. "As a result, they only provide relative temperature maps," Warren added. "So we're developing methods to do MRI differently."
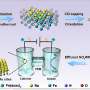
The Duke group's approach involves selective detection of what are called "intermolecular multiple quantum coherences (iMQCs)" in hydrogen atoms. Warren said the use of iMQCs is an application of his lab's 1998 correction of an early "subtle mistake" in the way MRI's inventors exploited quantum mechanical theory
While MRI theory sees nuclei of hydrogen as miniscule bar magnets spinning in characteristic ways within magnetic fields, it originally ignored certain interactions between those spins, Warren said. "We had to completely rewrite the theory of magnetic resonance to figure out where the mistake was made," he added.
By incorporating these missing interactions, the Duke chemists refinied both the electronics and interpretation of data from MRI scans to improve heat measurements.
The Duke method exploits three sets of facts: First, water and fat never mix. Secondly, hydrogen atoms in water respond to heat changes but those in fat don't. Thirdly, water and fat molecules in the body are likely to be positioned within tens of millionths of a meter (or microns) of each other.
Fat and water molecules occurring so close together are subjected to the same magnetic conditions, the Duke chemists reasoned. So the differences between the two types of MRI signals they emit should represent the effect of temperature changes on the hydrogen in water. Calculating the effects of iMQCs -- the subtle interactions between atomic spins -- further improves the accuracy of the comparison.
"So the difference between water and fat is an absolute magnetic resonance thermometer," Warren said.
The Duke team's report notes that the technique has been demonstrated in live rodents, including obese animals whose cells mimic those in fatty breast tissue. Because of fat cells' effects on magnetic fields, breast tissue cannot be temperature-checked using conventional MRI, the report also noted.
The technique could improve clinical applications of hyperthermia against cancer, and also be applied in other kinds of therapy, Warren suggested. "Temperature regulation is an extremely important part of how biological processes in us work," he said.
Source: Duke University