Novel structure proteins could play a role in apoptosis
Isoforms from Novel Structure Proteins (NSP), a new family of genes discovered by researchers in the Sbarro Institute for Cancer Research and Molecular Medicine in Temple University's College of Science and Technology, could be involved in apoptosis or programmed cell death.
NSPs were discovered four years ago by Nianli Sang, then a doctoral student at the University and now an assistant professor at Thomas Jefferson University and the Cardeza Foundation, and Antonio Giordano, director of the Sbarro Institute at Temple.
The researchers noted at that time that this family of genes sits mostly in the nucleus of our cells and exhibits the characteristics of a tumor-promoting gene. One form of the gene, the isoform NSP5a3a, is highly expressed in some tumor cell lines and could be very useful as a tumor marker, Giordano said. A protein isoform is a version of a protein with only small differences to another isoform of the same protein.
In the latest study, "Possible Functional Roles of NSPs in Cancer," published in Cell Cycle (Vol. 7, Iss. 12), the Temple researchers examined the gene in Hela cells, or cervical cancer cell lines, to further characterize the possible role of these NSPs.
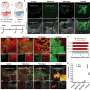
"So far, we've been able to confirm that two of the NSPs' four isoforms, called NSP 5a3a and NSP 5a3b, interact with a protein called B23, a multifunctional protein that is involved in cellular processes such as cell division, DNA repair and apoptosis," said Luca D'Agostino, a research fellow in the Sbarro Institute and the study's lead author.
In the study, the researchers silenced the expression first of NSP 5a3a. They also did the same for B23, and both in combination, always in-vitro.
"We saw that when we lowered the expression of NSP 5a3a, the cell's viability was not affected and they continued proliferating," D'Agostino said. "They must have a mechanism to compensate for its absence."
But when the expression of B23 was lowered, most of the cells moved towards apoptosis, which has already been established in previous studies, he said.
When the expression of both proteins was lowered simultaneously, D'Agostino noted, no apoptosis occurred, leading the researchers to conclude that at least in this model of in-vitro Hela cells, NSP 5a3a, working in association with B23, may have a role in apoptosis.
"Since these NSP isoforms have similar structural characteristics of known structural proteins such as spectrins, they may act as scaffolding proteins to help B23 interact with whoever it needs to interact with inside the cells," he said. A spectrin is a cytoskeletal protein that lines the intracellular side of the plasma membrane of many cell types in pentagonal or hexagonal arrangements, forming scaffolding that plays an important role in maintenance of plasma membrane integrity and cytoskeletal structure.
D'Agostino said the next step for researchers is to examine exactly how the NSP isoforms are interacting with the B23 protein. "We have an idea, but we still have to confirm it," he said.
D'Agostino added that these NSP isoforms may also have diagnostic value.
"Eventually, we have to screen different types of cancers, different grades and stages, to see if there is a difference in the expression of NSP5a3a and 5a3b, and if they have an application in the clinical field," he said. "With the 5a3a, if we see a pattern of expression when we do screenings of tissue biopsies, it could be useful as a diagnostic tool."
Source: Temple University