'Smothered' genes combine with mutations to yield poor outcome in cancer patients
Johns Hopkins Kimmel Cancer Center researchers have identified a set of genes in breast and colon cancers with a deadly combination of traditional mutations and "smothered" gene activity that may result in poor outcomes for patients.
The Hopkins team showed that this smothering process, called epigenetic inactivation, contributes to the aggressiveness of breast and colon cancer by disrupting biochemical pathways that normally suppress the runaway growth of cells that is the hallmark of cancer. While mutations alter pathways by rewriting the gene's DNA code, epigenetic marks affect genes without changing the code itself.
"Until studies like ours, it was easy to think that if we didn't find gene mutations in certain biochemical pathways linked to breast or colon cancer, then those pathways were normal in such patients," says Stephen Baylin, M.D., the Virginia and D.K. Ludwig Professor for Cancer Research and deputy director of the Kimmel Cancer Center. "Now we know that, in some patients, the pathways involved with newly discovered mutated genes are often more frequently disrupted by epigenetic mechanisms rather than genetic ones."
"That's a powerful insight that could help us diagnose patients quicker, predict the course of their cancer more accurately and in the future treat the disease more effectively," adds Baylin. A report on this work appeared May 27 in PLoS Medicine.
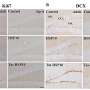
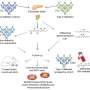
The team made their discovery using microarray technology - special silicon chips carrying pieces of genetic material that allow thousands of genes to be analyzed at one time. For this study, microarrays were tailored to locate cancer-related genes inactivated by an epigenetic process called DNA methylation. This methylation involves the binding of molecules called methyl groups to elements of DNA called cytosines that are located in a gene's "on-off switch." Excess methylation smothers the gene with too many methyl groups and interferes with the gene's normal protein production, setting the stage for a lethal cancer.
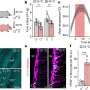
Some 189 mutated genes in breast and colon cancers, previously identified by a Kimmel Cancer Center research team, were screened for methylation by Baylin's group. They found 36 genes that were infrequently mutated in cancer, but were "hyper"methylated, often in both breast and colon cancers. After reviewing samples from 30 breast and 20 colorectal cancer patients as well as information from public microarray databases, the researchers found 18 of these genes that were strongly linked to poor outcome of patients with tumors carrying these changes.
For most of the genes, the researchers were able to reverse their epigenetic change and reactivate them in test tubes by stripping off excess methyl groups. This suggests that new treatments designed to reverse hypermethylation could be a simpler and more practical approach to treating cancer than strategies that attempt to replace, deactivate or compensate for mutated genes, according to Baylin.
Baylin also believes that the methlylated genes identified in this study could be inactivated in a broader range of cancers as well. That means the current findings could be extended to other cancers, improving the ability of physicians to predict the course of additional types of tumors, he says.
"We've learned from this study that we must include both genetic and epigenetic changes when we do future microarray analyses to increase our understanding of the genetic basis of cancer," Baylin says. "Such information will provide new details about why cancers start and help us identify which cancers will be particularly aggressive in our patients."
Provided by Johns Hopkins Medical Institutions