Researchers find targeted therapy combination overcomes treatment resistance in liver cancer
Researchers at the University of Pennsylvania School of Medicine and Abramson Cancer Center reported today at the annual meeting of the American Association for Cancer Research that combining two targeted therapies overcomes treatment resistance in liver cancer cell lines. The team is currently designing a trial to test the combination in patients.
Liver cancer is resistant to many chemotherapies and to cell-death inducing agents. Last year, however, the U.S. Food and Drug Administration approved sorafenib (Nexavar®) as a treatment for liver cancer after a clinical trial showed that the targeted agent prolonged survival in some patients.
Unfortunately not all patients respond to sorafenib and the drug does not cure the disease.
Therefore, Wafik El-Deiry, MD, PhD, Professor of Medicine, Genetics, and Pharmacology, and co-Program Leader of Radiation Biology in the Abramson Cancer Center, and colleagues have tested other targeted agents in combination with sorafenib.
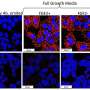
They found that treating liver cancer cells with sorafenib and an antibody or the natural ligand that stimulates programmed cell death via the TRAIL pathway, dramatically increases the rate of cell death.
“Sorafenib by itself causes a little cell death, but not that much,” Dr. El-Deiry said. “Now you combine sorafenib and TRAIL, and all of the sudden you get massive cell death. It is a real synergistic interaction. It is very profound killing.”
The combination works regardless of whether the researchers use a monoclonal antibody that stimulates the TRAIL receptor, which resides on the surface of the cancer cells, or the receptor’s natural ligand, a small protein called TRAIL. Both the antibody and the TRAIL ligand are currently being tested as single agents in patients.
Within the next several months, Dr. El-Deiry’s team expects to announce the details for a trial testing the combination in patients.
In a healthy individual, the immune system uses the TRAIL pathway to rid the body of unwanted cells, including precancerous ones. Once cancer develops, however, the cells often become less responsive to TRAIL activation, in part because of an overabundance of a protein called Mcl-1, according to Dr. El-Deiry. His team found that sorafenib reduces the amount of Mcl-1 in the cancer cells, restoring their sensitivity to TRAIL-induced cell death.
Although the Penn investigators focused their current report on liver cancer, they discovered that the sorafenib-TRAIL combination also kills colon cancer cell in vitro and in animal models.
Source: University of Pennsylvania