New regulatory circuit identified for aggressive, malignant brain tumor
Research using a newly developed algorithm has significantly advanced understanding of the molecular events associated with the most common primary brain tumor in adults, human glioblastoma (GBM). The research, published by Cell Press in the April issue of the journal Cancer Cell, validates known genetic aberrations and identifies events not previously linked with GBM, thereby elucidating new directions for potential therapeutic strategies.
GBM is a devastating neurological cancer that is characterized by widespread invasion, robust angiogenesis and a stubborn resistance to conventional and targeted treatments. Previous research has implicated a long and highly complex list of genes in GBM pathogenesis, emphasizing the need for a systemic prioritization that would separate relevant target genes from bystanders and provide detailed knowledge of the complex interactions between multiple disrupted genes and downstream targets.
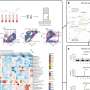
Dr. Lynda Chin from Dana-Farber Cancer Institute, Dr. Cameron Brennan from Memorial Sloan-Kettering Cancer Center and their colleagues used a high-resolution genome topography scan (GTS) algorithm to both define and rank the patterns of genomic alterations associated with GBM in primary samples and cell lines. Using this new methodology, the researchers discovered an unanticipated co-deletion pattern among closely related INK genes in the GBM oncogenome.
Specifically, the researchers identified a frequent co-deletion of p18INK4C and p16INK4A, a pattern unique to glioblastoma. Functional reconstitution of p18INK4C in GBM cells lacking both p16INK4A and p18INK4C resulted in impaired cell cycle progression and tumorigenic progression. Depletion of p18INK4C in p16INK4A-deficient primary glial cells or established GBM cells enhanced tumorigenicity while acute suppression of p16INK4A in primary glial cells induced an increase in p18INK4C. “These results uncover an unanticipated tumor suppressor role for p18INK4C in human GBM wherein it functions cooperatively with other INK4 family members to constrain inappropriate glial cell proliferation,” concludes Dr. Chin.
“This study also demonstrates that GTS can address one critical need in the development of a functional map of GBM genetic targets: namely, to prioritize those genomic alterations that are likely to be of importance from among those that are more likely to be bystanders of the cancer process,” offers Dr.Brennan. “Downstream functional validation of high probability candidates should yield novel GBM genes and potential targets for therapeutic intervention.”
Source: Cell Press