Epidemic superbug strains evolved from one bacterium: study
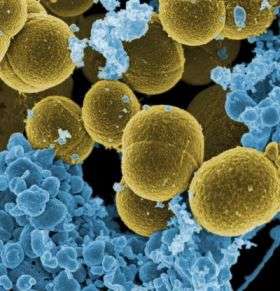
Community-associated methicillin-resistant Staphylococcus aureus (CA-MRSA) infections are caused primarily by a single strain—USA300—of an evolving bacterium that has spread with “extraordinary transmissibility” throughout the United States during the past five years, according to a new study led by National Institutes of Health (NIH) scientists. CA-MRSA, an emerging public health concern, typically causes readily treatable soft-tissue infections such as boils, but also can lead to life-threatening conditions that are difficult to treat.
The study, from the National Institute of Allergy and Infectious Diseases (NIAID) of NIH, resolves debate about the molecular evolution of CA-MRSA in the United States. The findings rule out the previously held possibility that multiple strains of USA300, the most troublesome type of CA-MRSA in the United States, emerged randomly with similar characteristics. The study also offers a hypothesis for the origin of previous S. aureus outbreaks, such as those caused by penicillin-resistant strains in the 1950s and 1960s.
A second study led by the same NIAID scientists takes the issue of the evolution of MRSA a step further, revealing new information about how MRSA bacteria in general, including the USA300 group, elude the human immune system.
The first study, which appears online this week in the Proceedings of the National Academy of Sciences, found that the USA300 group of CA-MRSA strains, collectively called the epidemic strain, comprises nearly identical clones that have emerged from a single bacterial strain. It is the first time scientists have used comparative genome sequencing to reveal the origins of epidemic CA-MRSA. Frank R. DeLeo, Ph.D., at NIAID’s Rocky Mountain Laboratories (RML) in Hamilton, Mont., led the research.
“Scientists are pressing ahead quickly to learn more about how some MRSA strains evade the immune system and spread rapidly,” says NIAID Director Anthony S. Fauci, M.D. “The information presented in these two studies adds important new insights to that expanding knowledge base.”
To understand how CA-MRSA is evolving in complexity and spreading geographically, Dr. DeLeo’s group sequenced the genomes of 10 patient samples of the USA300 bacterium recovered from individuals treated at different U.S. locations between 2002 and 2005. They then compared these genomes to each other and to a baseline USA300 strain used in earlier studies. Eight of the 10 USA300 patient samples were found to have nearly indistinguishable genomes, indicating they originated from a common strain. The remaining two bacteria were related to the other eight, but more distantly.
Interestingly, of the eight nearly indistinguishable USA300 patient samples, two caused far fewer deaths in laboratory mice than the others, highlighting an emerging view that tiny genetic changes among evolving strains can profoundly affect disease severity and the potential for drug resistance to develop.
“The USA300 group of strains appears to have extraordinary transmissibility and fitness,” says Dr. DeLeo. “We anticipate that new USA300 derivatives will emerge within the next several years and that these strains will have a wide range of disease-causing potential.” Ultimately, Dr. DeLeo and his colleagues hope that the work will lead to the development of new diagnostic tests that can quickly identify specific strains of MRSA.
Fred C. Tenover, Ph.D., of the Centers for Disease Control and Prevention in Atlanta (CDC) contributed the 10 USA300 clinical isolates from CDC’s Active Bacterial Core Surveillance system. Other study collaborators included Barry N. Kreiswirth, Ph.D., of the International Center for Public Health (ICPH) in Newark, N.J., and James M. Musser, M.D., Ph.D., of The Methodist Hospital Research Institute in Houston.
The second report, which involved scientists from RML, ICPH and Vanderbilt University Medical Center in Nashville, was recently published online in the Journal of Immunology. This study provides scientists with new details about the complex mechanisms MRSA uses to avoid destruction by neutrophils, human white blood cells that ingest and destroy microbes. When exposed to hydrogen peroxide, hypochlorous acid (the active component of household bleach) or antimicrobial proteins—all killer chemicals released by neutrophils—MRSA senses danger, escapes harm and turns the tables on the white blood cells, destroying them. Work is continuing in Dr. DeLeo’s lab to understand how the bacterium senses and survives attacks by neutrophils.
References: A Kennedy et al. Epidemic community-associated methicillin-resistant Staphylococcus aureus: Recent clonal expansion and diversification. Proceedings of the National Academy of Sciences DOI:10.1073/PNAS.0710217105 (2008).
Source: National Institute of Allergy and Infectious Diseases