Alzheimer's molecule is a smart speed bump on the nerve-cell transport highway
Researchers at the University of Pennsylvania School of Medicine discovered that proteins carrying chemical cargo in nerve cells react differently when exposed to the tau protein, which plays an important role in Alzheimer’s disease.
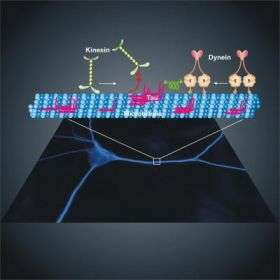
Dynein and kinesin proteins transport cellular cargo towards opposite ends of tracks called microtubules. Tau binds to the microtubule surface and acts like a speed bump to regulate protein traffic, the group found. “But it is a smart speed bump because it impedes these different motor proteins to different degrees,” explains first author Ram Dixit, PhD, a postdoctoral fellow in the lab of senior author Erika Holzbaur, PhD, Professor of Physiology.
“Our findings show a mechanism of regulating the transport of nutrients, signaling molecules, and waste proteins along a nerve cell’s axon,” says Dixit. “Neurodegenerative diseases such as Alzheimer’s arise when pieces of this shipping system goes awry.”
The transport performed by dynein and kinesin is required for continuously providing new proteins to the axon and synapse to maintain normal cellular function, and is also required to remove old, misfolded, or aggregated proteins for degradation. Just as important, this transport is required for moving other proteins from the nerve-cell synapse back to the cell body, which is also required to maintain healthy neurons.
In neurons, microtubules are abundantly decorated with tau. Dynein and kinesin encounter the tau molecules on their travels along the microtubules. The Penn group found that dynein, which carries loads towards the interior of the cell, maneuvers around tau; whereas, kinesin, which carries loads towards the outside of the cell, detaches when it encounters tau.
These findings appear online January 17 in Science in advance of print publication.
Dynein and kinesin’s individual maneuverings when encountering tau allow for the cell to be able to offload cargo where it needs to go with fine-tune accuracy. “Tau may determine where kinesin offloads cargo along the microtubule tracks that radiate out to the cell surface from the center,” says Holzbaur. “And dynein’s ability to back up and go around when it encounters an obstacle such as tau may be a mechanism to ensure that it gets to the center of the cell with its important cargo.”
The group conducted studies using single molecules moving along a tau-decorated microtubule to determine the effects of tau on dynein and kinesin’s movement. Mutations in molecular motors such as dynein and kinesin can lead to degeneration of neurons. These mutations, for example, decrease the efficiency of dynein and kinesin. This problem can lead to the accumulation of misfolded proteins in the cell, which, in turn may lead to the degeneration of the neuron.
“There’s a growing theme that defects in transport are tightly associated with neurodegeneration,” says Holzbaur. “It’s already been shown in Alzheimer’s disease that there is a change in the distribution of tau along microtubules. Instead of kinesin getting its cargo closer to the cell’s outer surface, tau accumulates in the cell body and kinesin’s cargo of newly synthesized proteins gets dropped early, not at the cell surface, thus causing problems. Our findings explain how that can happen.”
Source: University of Pennsylvania