A novel way found to prevent protein plaques implicated in Alzheimer's
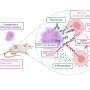
For unknown reasons a protein called amyloid beta aggregates into toxic plaques in the brain, killing neurons. These plaques are one of the hallmarks of Alzheimer’s disease. Now two new animal studies show for the first time that the deadly transformation of amyloid beta into plaques can be prevented through an interaction between amyloid beta and another protein called cystatin C.
Although much work needs to be done, these new findings may open the door to new treatments for Alzheimer’s disease that mimic the effects of cystatin C, says Efrat Levy, Ph.D., Associate Professor in the Departments of Psychiatry and Pharmacology at New York University School of Medicine, and the lead author of the study.
“We are really excited by these findings because recent studies show that cystatin C is protective against a variety of insults that cause cell death in the brain. Our potential therapeutic approach focuses on keeping amyloid beta in a water soluble form, preventing its accumulation in the brain, and thus slowing, halting, or reversing disease progression,” says Dr. Levy, who is also Director of the Laboratory of Molecular Pathology of Cerebral Amyloidosis at the Nathan S. Kline Institute for Psychiatric Research in Orangeburg, New York.
With support from the Alzheimer’s Association, Dr. Levy’s laboratory has already begun studies to develop a drug that will mimic the ability of cystatin C to bind to a non-pathological form of amyloid beta and thereby prevent its accumulation into plaques in the brain.
Alzheimer’s is estimated to affect 5 million Americans and there are no medicines that can delay or prevent the disease. Many laboratories worldwide are pursuing ways to prevent the clumping of amyloid beta as possible therapies for the disease. It isn’t known whether the protein actually causes Alzheimer’s, but amyloid beta is one of the proteins implicated in the disease process.
The two studies appear in the on-line November 18, 2007, edition of the journal Nature Genetics and will be published in its December 2007 issue.
The first study was performed by Dr. Levy in collaboration with other investigators at the Nathan S. Kline Institute for Psychiatric Research and New York University School of Medicine. The second study was performed by Dr. Levy and colleagues in the laboratory of Dr. Mathias Jucker at the Hertie-Institute for Clinical Brain Research in Tubingen, Germany.
Both studies used mice that were genetically engineered to produce human cystatin C as well as abundant amounts of amyloid beta plaques in their brains. The cystatin C bound to the soluble, non-pathological form of amyloid beta in these mice and inhibited the aggregation and deposition of amyloid beta plaques in the brain.
The research shows that cystatin C binds soluble amyloid beta also in the human brain, and suggests that this binding inhibits its aggregation into insoluble plaques in humans, says Dr. Levy. Cystatin C production and body fluid levels vary among healthy individuals and can be influenced by certain hormones, aging, and certain pathological conditions, she says. Furthermore, it was recently demonstrated that a genetic variation in the cystatin C gene in human populations is linked to a greater risk of developing Alzheimer’s disease during aging.
These findings suggest, says Dr .Levy, that even subtle modifications of cystatin C protein levels could affect amyloid beta accumulation and deposition in the brain, thereby modifying disease progression.
Cystatin C is found in all body fluids and tissues of mammals. It is a jack-of-all-trades, playing a broad role in many functions, including cell proliferation and growth, modulation of the inflammatory response, and bone resorption. The protein also has been implicated in neuronal degeneration and repair of the nervous system.
“The work of Dr. Levy and colleagues offers important insights into how cystatin C may lower the risk for Alzheimer’s disease, setting the stage for a new approach to therapy,” said Ralph A. Nixon, M.D., Ph.D., Professor of Psychiatry and Cell Biology at NYU School of Medicine and Director of the Center for Dementia Research at the Nathan S. Kline Institute. Dr. Nixon also directs the Silberstein Institute at NYU School of Medicine.
“Given the increasing prevalence of this disease, its staggering social and economic costs for patients and their families, and the limitations of existing therapies, we are very hopeful that this research will lead to improved medications to prevent the disease and forestall its devastating symptoms,” said Dr. Harold S. Koplewicz, Director of the Nathan S. Kline Institute, and the Chair of the Department of Child and Adolescent Psychiatry and a Vice-Dean at NYU School of Medicine.
Source: New York University