Researchers identify molecules with interesting anti-clotting properties
Virginia Commonwealth University researchers have discovered a new mechanism to inhibit key enzymes that play a major role in clotting disorders, which could lead to novel therapies to treat clots in the lungs and those localized deep in the body in areas such as the legs.
Antithrombotic disorders occur when the effect of thrombin, a protein involved in coagulation, is inhibited, rendering blood unable to clot effectively. These disorders are considered common and can be fatal. Additionally, clotting disorders arise due to complications from other diseases like cancer. Although there are a number of anticoagulation drugs available -- heparins and warfarins -- some patients develop adverse reactions to the therapy and must be closely monitored.
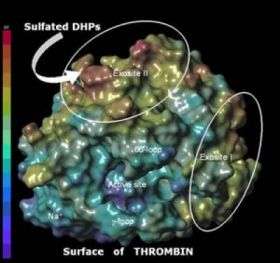
In a study published in the Nov. 2 issue of the Journal of Biological Chemistry, Umesh R. Desai, Ph.D., a professor in the Department of Medicinal Chemistry at the VCU School of Pharmacy, lead investigator on the study, reported on the design of three highly complex molecules with unique anticoagulant properties that were prepared in the laboratory. According Desai, these molecules, known as sulfated DHPs, are completely different from anticoagulants used in the clinic today including heparins, coumarins and hirudins.
The team demonstrated that the molecules were able to inhibit the ability of critical enzymes involved with the cascade of events involved in blood clotting. Specifically, the molecules prevent the normal action of thrombin and factor Xa, which are the critical enzymes targeted by current anticoagulant therapy.
“We have identified a new mechanism that may prevent clotting. This approach may result in new drugs for the treatment of thrombotic disorders, including pulmonary embolism, deep vein thrombosis and more,” said Desai.
“The molecules we have designed may possess several advantages compared to currently available anticoagulation drugs,” he said.
“For example, new anti-clotting therapies may result in reduced hospital stays for patients, fewer side effects, and possibly an overall cost reduction in therapy because our molecules are likely to be synthesized in an inexpensive manner.”
Desai and his team are now investigating which unit or units in the complex molecule are responsible for the anti-clotting activity.
Source: Virginia Commonwealth University

















