Persistence pays off
Chemists have come remarkably close to mimicking a type of protein previously thought impossible to imitate. The long-term application of this work could be in the development of new types of glucose sensors for diabetics, or in the production of new biofuels.
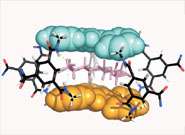
Professor Anthony Davis and his team at the University of Bristol, UK, report this week in Science that they have designed a totally synthetic molecule which mimics the properties of lectins. The molecule binds a particular kind of carbohydrate (cellobiose), while ignoring almost all other carbohydrates. This has previously proved extremely difficult to do.
Lectins are proteins which bind to carbohydrates such as glucose. They are found throughout nature, particularly in plants. By designing synthetic molecules with appropriate shapes, chemists should, in theory, be able to mimic the properties of such biomolecules and then adjust them to suit the needs of humanity. Unfortunately this has proved extremely difficult in practice.
Lectins are large, complex proteins which have evolved to do their jobs over billions of years and it is generally assumed that we cannot realistically hope to match them with artificial systems. Indeed, efforts to do so have usually been disappointing. But research at Bristol University suggests that perhaps we shouldn't give up so easily.
Professor Davis said: “This work should help scientists understand lectins themselves and may lead to specific applications in the future. For example, cellobiose is closely related to cellulose, a key renewable resource which can be used to make materials, biofuels and other chemicals.
“Cellulose is difficult to exploit because of its insolubility, but if it could be bound and made soluble by a synthetic lectin, new ways of processing it should be possible. The researchers are also aiming for molecules which bind selectively to glucose. These could be used in new types of glucose sensor, for use in the treatment of diabetics.”
Source: University of Bristol















