What is the lifetime of positronium ions?
The positronium ion is the most simply built negative ion imaginable, made of just two electrons and a positron. This exotic combination is unstable: after just a few fractions of a billionth of a second it decays into gamma rays. Scientists from the Max Planck Institute for Nuclear Physics in Heidelberg have now begun closely examining this short-lived three-particle system. With an unprecedented accuracy, they measured the lifetime of positronium ions.
The researchers are hoping to gain a better understanding of quantum-mechanical many-body systems; the measurements are just the beginning of a longer-term project investigating positronium ions.

All naturally-occurring chemical elements have a similar basic structure: the atomic nucleus contains protons and neutrons, and around it are electron shells. But exotic atoms, like positronium, are different. It is made of an electron and its anti-particle, the positron. Positrons have exactly the same characteristics of an electron, except for an opposite electrical charge. When positrons and electrons come together, they annihilate each other very quickly. The entire rest mass of positronium is thereby converted into gamma rays. For this reason, positronium atoms exist only for fractions of a billionth of a second, and they are rarely observed in nature. Whenever they are created, they quickly disappear.
Physically, however, the structure of positronium atoms is simple to explain. They are a system of two practically identical particles that are attracted to each other with only a single force: "electroweak interaction". (under normal conditions, even the electromagnetic force is generally enough. On the other hand, in "typical" atoms made from neutrons, protons, and electrons, the spatial size of the nucleus, and additional strong interaction, complicate the situation.)
Things become even more interesting when a further electron is attached to the positronium atom. In this kind of negatively-charged atom (in other words, an ion), the three particles interact with each other. Understanding how this works is a challenge for physicists, because the characteristics of three-body systems can only be approximated. The positronium ion provides an interesting model system for quantum mechanics, because of its simplicity.
Until now there has not been the required experimental data to test the approximation procedures used in theory. The first thing that Max Planck scientists did in their project investigating positronium ions was to measure the lifetime of the exotic atoms six times more precisely than had ever been done before. The scientists found that positron ions live on average for almost half a nanosecond (0.4787 ns). This closely matches the predicted lifetime.
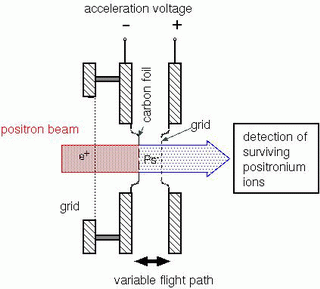
The measurements lasted eight months. The researchers fired positrons through a very thin carbon foil. About every 10,000th positron became attached to two electrons, creating a positronium ion. Because their lifetimes are too short for a direct measurement, it must be done indirectly. In order to do this, the ions are accelerated in an electrical field at velocities up to 7% the speed of light, and sent over a very precise, adjustable flight path (Figures 1 and 2). The lifetime can then be determined by counting the number of positronium ions arriving at the other end, at various distances. At these speeds, the range of the ions is of the order of millimetres - a practical, easily manageable scale.
The researchers have further experiments planned for the future. With the launch of the positron source NEPOMUC at the research reactor FRM-II near Munich, Germany, there will be a high-intensity positron beam available. It will enable an even more precise measurement of the lifetime, and other characteristics, of this unusual ion.
Original work:
Frank Fleischer, Kai Degreif, Gerald Gwinner, Michael Lestinsky, Vitaly Liechtenstein, Florian Plenge, Dirk Schwalm
Measurement of the decay rate of the negative ion of positronium
Physical Review Letters, February 13, 2006 (online)
Source: Max Planck Institute for Nuclear Physics
















