MIT invents 'lab on a chip' to automate gene studies
Genetic studies on whole animals can now be done dramatically faster using a new microfluidic chip developed by engineers at MIT.
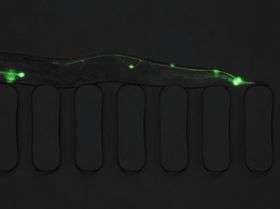
The new "lab on a chip" can automatically treat, sort and image small animals like the 1-millimeter C. elegans worm, accelerating research and eliminating human error, said Mehmet Yanik, MIT assistant professor of electrical engineering and computer science.
Yanik and his colleagues described their device in the advance online issue of the Proceedings of the National Academy of Sciences the week of Aug. 20. "Lab on a chip" technologies are being developed to sort and image individual cells, but this is the first device that can be used to study whole animals.
C. elegans is often used in studies designed to identify which genes control which phenotypes, or traits. Researchers traditionally do this by treating them with a mutagen, or by using RNA interference, in which expression of a certain gene is blocked with a small strand of RNA. Such studies normally take months or years to complete. The new chip, which sorts and images worms in milliseconds, dramatically speeds up that process.
"Normally you would treat the animals with the chemicals, look at them under the microscope, one at a time, and then transfer them," Yanik said. "With this chip, we can completely automate that process."
The tiny worms are flowed inside the chip, immobilized by suction and imaged with a high resolution microscope. Once the phenotype is identified, the animals are routed to the appropriate section of the chip for further screening.
The worms can be treated with mutagen, RNAi or drugs before they enter the chip, or they can be treated directly on the chip, using a new, efficient delivery system that loads chemicals from the wells of a microplate into the chip.
"Our technique allows you to transfer the animals into the chip and treat each one with a different gene silencer or a different drug," Yanik said.
Yanik and his colleagues plan to use the chips to continue their research on neural degeneration and regeneration in C. elegans. Yanik and his collaborators had previously demonstrated a high precision femtosecond laser technology to cut axons in living animals and then observe which genes are involved in axon regeneration.
The lead author of the paper is Chris Rohde, a graduate student in electrical engineering and computer science (EECS). Other authors of the paper are Matthew Angel, a graduate student in EECS, Fei Zeng, a postdoctoral fellow in the Research Laboratory of Electronics, and Ricardo Gonzalez-Rubio, a graduate student in biological engineering.
Source: MIT