Potential new target for cancer found
By bypassing a well-known gene implicated in almost one-third of all cancers and instead focusing on the protein activated by the gene, Duke University Medical Center researchers believe they may have found a new target for anti-cancer drugs.
In experiments with human cells and animal models, the researchers studied the gene known as “Ras,” which is integral in normal cell growth. When this gene is mutated and becomes overactive, it can lead to the unregulated proliferation of cells that is the hallmark of tumor formation.
The ras gene, known as an oncogene when it is in this mutated state, has been implicated in several different cancers, including those of the pancreas and lungs. To date, efforts at blocking or turning off ras have proven ineffective. Pancreatic cancer has been shown to have the strongest link to the ras oncogene, and it is also one of the hardest cancers to treat, with few patients alive five years after diagnosis, researchers said.
“Since it has been so difficult to target the ras gene itself with drugs, we tried to determine if something that ras activates could be a possible target for a drug or therapy,” said Christopher Counter, Ph.D., associate professor of pharmacology and cancer biology and senior member of the research team. “We found a specific target that could be susceptible to drugs, and if these findings are proven true in human trials, we could have a new way of treating ras-dependent cancers.”
The results of the Duke experiments were published July 15, 2007, in the journal Genes & Development. Brooke Ancrile, a graduate student in Counter’s laboratory, was first author of the paper. The research was supported by the National Institutes of Health.
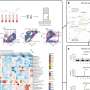
The researchers discovered that the overactive ras gene was responsible for above-normal secretion of a factor known as interleukin-6 (IL-6). Scientists know a great deal about IL-6 and its functions in the body, but its link to oncogenic ras was unknown.
In addition to finding that the ras oncogene spurred the production of IL-6, they also found that inhibiting IL-6 production reduced the creation of new blood vessels, which are crucial for the development and nourishment of tumors.
“IL-6 was like the gas pedal driving the growth of tumors,” Counter said. “No gas, no growth, which is exactly what we saw when we inhibited IL-6 in tumors.”
Counter is encouraged that even though these findings are in cell culture and animal models, therapies based on targeting IL-6 in cancers driven by the ras oncogene could be tested in humans in the near future. A biotechnology company has already developed a monoclonal antibody specific to IL-6 which could be used to neutralize IL-6.
A phase II trial is underway testing a monoclonal antibody against IL-6 for patients with multiple myeloma, a cancer that depends on IL-6 but is not known to have a connection to the ras oncogene. If the results of this trial are positive, studies might begin in ras-dependent cancers. Counter’s group is actively pursuing the idea that such an antibody may inhibit pancreatic cancer growth in mouse models. If these results are positive, this will open the door for Duke oncologists to organize a clinical trial to test the agent in human cancer patients.
“Secreted proteins promoting the growth of blood vessels in tumors have been successfully neutralized in the past with antibodies,” Ancrile said. “We believe that IL-6 is a viable target for drugs that holds promise in the treatment of cancers dependent on the ras oncogene.”
Source: Duke University Medical Center