Targeting key proteins of carcinogenesis
Misfolded and disused proteins are eliminated by a cellular shredder called the proteasome. The cell labels the proteins it wants to dispose with Ubiquitin (Ub) in order to avoid the unwanted degradation of still needed proteins. Malfunctions in the ubiquitin-proteasome system can be fatal for the organism.
In particular cancer and immunological disorders but also developmental defects are the consequences. The knowledge about the ubiquitination reaction as well as the effect of ubiquitination on diverse cellular functions has already led to the development of highly promising drugs like Bortezomib and Herceptin. The molecular mechanism of Ub conjugation and its biological meaning has been discovered by Avram Hershko, Aaron Ciechanover and Irwin Rose who have been awarded the noble price for their work in 2004.
In the current issue of Molecular Cell Prof. Ivan Dikic and his team from the institute of biochemistry II at the University of Frankfurt now describe a novel Ub conjugation reaction that might allow a more efficient manipulation of key proteins in the treatment of cancer and other diseases. “Our current view on protein ubiquitination as it is found in the textbooks has to be revised”, explains Prof. Müller-Esterl, director of the institute of biochemistry and vice president of the Johann-Wolfgang-Goethe University.
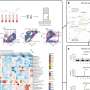
“Virtually every process within an eukaryotic cell is directly or indirectly controlled by ubiquitin”, says Dikic. This includes fundamental cellular programs such as protein degradation, DNA replication, signal transduction or protein trafficking. Up to now it was believed that the cooperation of three enzymes is needed to attach Ub to another protein: E1, E2 and E3. E1 activates Ub and transfers it to E2, which in turn cooperates with an so-called Ub ligase (E3) that couples Ub to a specific target protein. However, Dikic and colleagues discovered that certain proteins can be ubiquitinated independently of E3 ligases which caused a stir among experts.
A prerequisite for the E3-independent reaction is the presence of a Ub-binding domain (UBD) in the protein that is able to recognize Ub. “This type of protein is of utmost importance for the cell because it can distinguish between ubiquitinated and non-ubiquitinated proteins”, explains Dr. Daniela Hoeller, the responsible scientist. UBD-proteins are the key to the effects of Ub – both in normal and malignant cells.
Dikic and his coworkers demonstrated that proteins equipped with an UBD can ubiquitinate themselves by directly recruiting Ub-loaded E2 enzymes thus making E3 ligases dispensible. In their previous work Hoeller and Dikic showed that the ubiquitination of UBD proteins does not lead to their degradation but rather causes their functional inactivation. In other words they become unable to recognize and control ubiquitinated proteins. If needed the cell can re-activated them by cleaving the attached Ub. In this way the cell can quickly and dynamically react on external signals.
The discovery provides the basis for novel therapeutic approaches that are more specific than drugs like Bortezomib. “For an effective therapeutic use there is still a long way to go”, explains Müller-Esterl, who needs to update his biochemistry textbook now.
Source: Johann Wolfgang Goethe-Universität Frankfurt