Paving the way toward a vaccine against Alzheimer's disease
Scientists have provided new details about how proteins used to destroy bacteria and viruses may help treat Alzheimer’s disease. Gunnar K. Gouras, associate professor of neurology and neuroscience at Weill Medical College of Cornell University, New York, and colleagues provide new insights into how these proteins, called antibodies, reduce the main hallmarks of Alzheimer’s disease and raise hopes for a vaccine against the disease.
“Antibodies are probably the most promising experimental approach to fight Alzheimer’s disease at this time,” Gouras says. “The discoveries made using antibodies are so encouraging that results of ongoing vaccine trials against the disease are much anticipated.”
Alzheimer’s disease, the most common form of dementia, gradually destroys a person's memory and ability to learn, communicate, and carry out daily activities. According to the American Health Assistance Foundation, more than 4.5 million people in the United States live with the disease and more than 26 million people are affected worldwide. By 2050, the number of people who will suffer from the disease is estimated to nearly triple in the United States and to be four times as high worldwide.
Although no cure for the disease is available yet, scientists are actively looking for new treatments. One of the main goals of such treatments is to destroy clumps of a protein called beta amyloid, which are found in the brains of people with the disease, either inside the nerve cells or around them. Antibodies have been shown to be effective at removing these clumps but how they do it is not completely understood.
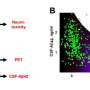
In their new study, appearing as the cover story of the June 29 issue of the Journal of Biological Chemistry, Gouras and his colleagues provide new details about how the antibodies attack these clumps inside the nerve cells. The study was selected as a “Paper of the Week” by the journal’s editor, meaning that it belongs to the top one percent of papers reviewed in significance and overall importance.
Using cultured cells from mice, the scientists showed that the antibodies first bind to the surface of the cells and connect to a protein called amyloid precursor protein (APP), which is already present on the cell surface. Then both proteins are internalized inside the cell.
Once inside the cell, APP is broken down into pieces, some of which are the amyloid beta proteins. If the antibodies are not present, the proteins start clustering and ultimately kill the cell. The scientists showed that the antibodies prevent this from happening by reducing accumulation of the amyloid beta proteins in vesicles inside the cell called endosomes.
“A lot of research has been done on protein clusters outside nerve cells,” Gouras says. “In this study, we investigated for the first time what happens inside the cells and how antibodies can help prevent clusters from forming.”
The researchers also found that the antibodies helped restore communication between nerve cells. In Alzheimer’s patients, the protein clusters alter parts of the cell surfaces – the synapses – that help nerve cells talk to one another. As a result, thoughts are not transmitted, memory is lost, and new learning is hindered. But Gouras and his team showed that the antibodies cleared the protein clusters and helped cells talk to one another again.
Over the past seven years, research results on the use of antibodies against Alzheimer’s disease have been so promising that two pharmaceutical companies, Ireland’s Elan Corp. and U.S. partner Wyeth, have been conducting clinical trials of a potential vaccine. Although the first trials were stopped when 6 percent of the patients developed encephalitis – an inflammation of brain tissue – other clinical tests on the treated patients have been encouraging. In the second half of this year, the two companies will test a potential drug, called Bapineuzumab, on patients with mild to moderate Alzheimer’s symptoms.
If successful, these trials could result in a new type of vaccine containing antibodies that would directly attack the amyloid beta protein clusters. Unlike common vaccines, which, in this case, would contain pieces of amyloid beta proteins and would stimulate the immune system to produce antibodies, the new vaccine would directly provide the antibodies to patients.
“These new developments are encouraging, but possible side effects may arise,” Gouras says. He adds that although clinical trials need to be conducted as soon as possible to help alleviate the suffering of the increasing number of Alzheimer’s patients, more research is still needed both to understand how amyloid beta proteins wreak havoc in the brain and to improve potential drugs.
Gouras and his colleagues are now trying to figure out how the protein clusters inside and outside the cells work together to destroy the cells. They already noticed that the clusters outside the cells affect those inside cells by making them grow. Another challenge will be to better understand what the clusters do inside the cells that leads to their death. The scientists are also using imaging and biochemical techniques to see, in cultured cells, how the antibodies affect the clusters.
“We have many indications that antibodies work,” Gouras says. “Now we need to understand how they do it.”
Source: American Society for Biochemistry and Molecular Biology